Summary
Leaf shape evolution in angiosperms is reviewed from an evolutionary developmental (evo/devo) viewpoint. Leaves have evolved as photosynthetic organs in land plants, while leaves in some angiosperms have lost their photosynthetic role, e.g., in some cacti, saprophytes/mycoheterotrophs, and parasitic plants. Although the roles are the same among leaves, their morphologies and developmental systems vary significantly, in part because of the need to maximize their photosynthetic efficiency for survival under particular environmental constraints. An example is the narrow leaves of the rheophyte plants located along river banks where frequent flooding occurs. Narrow leaves are less efficient at absorbing sunlight than are leaves with wider blades; however, they can withstand the destructive force of the water flow in full flood. In contrast, in some xerophytic epiphytes, the narrow leaves are effective at catching water from fog. Narrow leaf blade formation is also present in submerged amphibious plants, likely an adaptation to the underwater conditions. The leaf index, i.e., the ratio of leaf length to width, is regulated by several genetic factors, mutations of which may be driving the evolution of leaf shape. However, not all leaf shapes can be explained by environmental adaptation. For example, data on the relationship between leaf shape diversification and environmental habitats in terms of thermo regulation remain controversial.
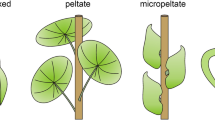
In contrast, understanding of the genetic/cellular mechanisms underlying leaf blade shape is increasing. Leaf blade organogenesis is governed by adaxial/abaxial identities, in which the plate meristem, responsible for flat leaf blade development, is activated along the interface between the adaxial and abaxial domains. Thus, some morphological diversity in leaves can be partly attributed to changes in the adaxial/abaxial patterning. For example, unifacial leaves, which have only an abaxial identity in the leaf blade, develop into stick-like or terete forms. They can stand vertically and thus live in densely populated areas, unlike plants with bifacial leaves which extend, in most cases, horizontally. Interestingly, some unifacial leaves are flat or ensiform, as seen in the genera Iris and Juncus. In such cases, since the plate meristem is not available, they rely on thickening growth to create flat lamina from the terete primordia. Lotus-like or peltate leaves are also derived from partial alterations in the adaxial/abaxial patterning; however, the pitcher leaves of carnivorous plants, once considered an extreme deformation of the peltate leaves, result from local alterations in cell division. In compound leaves, the major genetic regulatory components are shared with those of serrated leaf margins. The proportion of serrated-leaf species in any particular habitat precisely correlates with the average air temperature, and thus serration mechanisms are likely linked to physiological adaptations to the external environment. The evolution of determinate and indeterminate leaves in angiosperms is also discussed. While leaves are usually determinate and discarded periodically, some species develop indeterminate leaves, with some looking like twigs or lateral branches and functioning as twigs. Some are simple leaves and have an intermediate nature between a leaf and a shoot. We also see the evolution of leaf-like, photosynthetic organs. Some plants develop cladodes, which are modified lateral shoots that resemble leaves. Molecular analysis revealed that recruitment of dorsiventral control mechanisms in leaves has resulted in lateral shoots changing into leaf-like cladodes in the genus Asparagus. All of the above can be interpreted from an evo/devo viewpoint, but the question of why such great diversification occurred in angiosperm leaves remains.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- AN:
-
ANGUSTIFOLIA
- AN3:
-
ANGUSTIFOLIA3
- ARF:
-
AUXIN RESPONSE FACTOR
- AS1, AS2:
-
ASYMMETRIC LEAVES 1, ASYMMETRIC LEAVES 2
- ATGIF1:
-
Arabidopsis thaliana GIF1
- AtGRF:
-
Arabidopsis thaliana GRF
- BOP:
-
BLADE-ON-PETIOLE
- CIN-TCP:
-
CINCINNATA-class-TCP
- CRC:
-
CRABS CLAW
- CtBP/BARS:
-
C-terminal Binding Protein/ /BFA–ADP-Ribosylation Substrate
- CUC:
-
CUP-SHAPED COTYLEDON
- DL:
-
DROOPING LEAF
- ETT:
-
ETTIN
- FLO:
-
FLORICAULA
- evo/devo:
-
evolutionary developmental biology
- GA:
-
gibberellic acid
- GIF:
-
GRF-INTERACTING FACTOR
- GLK:
-
GOLDEN2-LIKE
- GRF:
-
GROWTH REGULATING FACTOR
- HD-ZipIII:
-
HOMEODOMAIN LEUCINE ZIPPER Class III
- KAN:
-
KANADI
- KNOX:
-
KNOTTED-LIKE HOMEOBOX
- KNOX I:
-
Class I KNOX
- L1:
-
Layer 1
- LFY:
-
LEAFY
- LNG:
-
LONGIFOLIA
- miR:
-
micro RNA
- NGA:
-
NGATHA
- oli:
-
oligocellula
- PIN1:
-
PIN-FORMED 1
- PHAN:
-
PHANTASTICA
- PRS:
-
PRESSED FLOWER
- RCO:
-
REDUCED COMPLEXITY
- ROT3:
-
ROTUNDIFOLIA 3
- RTFL/DVL:
-
ROTUNDOFOLIA FOUR-LIKE/DEVIL
- SAM:
-
shoot apical meristem
- STM:
-
SHOOT MERISTEMLESS
- tasiR:
-
trans-acting siRNA
- TCP:
-
TEOSINTE BRANCHED1, CYCLOIDEA, PROLIFERATING CELL NUCLEAR ANTIGEN FACTOR1 transcription factor
- WOX1:
-
WUSCHEL-LIKE HOMEODOMAIN 1
- xs:
-
extra-small sisters
- YAB:
-
YABBY
References
Adams WW III, Stewart J, Cohu CM, Muller O, Demmig-Adams B (2016) Habitat temperature and precipitation of Arabidopsis thaliana ecotypes determine the response of foliar vasculature, photosynthesis, and transpiration to growth temperature. Front Plant Sci 25:1026
Alvarez JP, Furumizu C, Efroni I, Eshed Y, Bowman JL (2016) Active suppression of a leaf meristem orchestrates determinate leaf growth. eLIFE 5:e15023
Andriankaja M, Dhondt S, De Bodt S, Vanhaeren H, Coppens F, De Milde L, Mühlenbock P, Skirycz A, Gonzalez N, Beemster GT, Inzé D (2012) Exit from proliferation during leaf development in Arabidopsis thaliana: a not-so-gradual process. Dev Cell 22:64–78
Bailey LW, Sinnott EW (1916) The climate distribution of certain types of angiosperm leaves. Am J Bot 3:24–39
Barkoulas M, Hay A, Kougioumoutzi E, Tsiantis M (2007) A developmental framework for dissected leaf formation in the Arabidopsis relative Cardamine hirsuta. Nat Genet 40:1136–1141
Bharathan G, Goliber TE, Moore C, Kessler S, Pham T, Sinha NR (2002) Homologies in leaf form inferred from KNOXI gene expression during development. Science 296:1858–1860
Bilsborough GD, Runions A, Barkoulas M, Jenkins HW, Hasson A, Glinha C, Laufs P, Hay A, Prusinkiewicz P, Tsiantis M (2011) Model for the regulation of Arabidopsis thaliana leaf margin development. Proc Natl Acad Sci U S A 108:3424–3429
Blein T, Pulido A, Vialette-Guiraud A, Nikovics K, Morin H, Hay A, Johansen IE, Tsiantis M, Laufs P (2008) A conserved molecular framework for compound leaf development. Science 322:1835–181839
Boyce CK (2007) Mechanisms of laminar growth in morphologically convergent leaves and flower petals. Int J Plant Sci 168:1151–1156
Brennan EB, Weinbaum SA, Rosenheim JA, Karban R (2001) Heteroblasty in Eucalyptus globulus (Myricales, Myricaceae) affects ovipositional and settling preferences of Ctenarytaina eucalypti and C. spatulata (Homoptera: Psyllidae). Environ Entomol 30:1144–1149
Chitwood DH, Headland LR, Ranjan A, Martinez CC, Braybrook SA, Koenig DP, Kuhlemeier C, Smith RS, Sinha N (2012) Leaf asymmetry as a developmental constraint imposed by auxin-dependent phyllotactic patterning. Plant Cell 24:2318–2327
Cronk QCB, Bateman RM, Hawkins JA (eds) (2002) Developmental genetics and plant evolution. Taylor & Francis, London/New York
de Reuille PB, Bohn-Courseau I, Ljung K, Morin H, Carrano N, Godin C, Traas J (2006) Computer simulations reveal properties of the cell-cell signaling network at the shoot apex in Arabidopsis. Proc Natl Acad Sci U S A 103:1627–1632
Dengler NG (1999) Anisophylly and dorsiventral shoot symmetry. Int J Plant Sci 160:S67–S80
Dengler NG, Dengler RE (1984) The mechanism of plication inception in palm leaves: Histogenic observations on the pinnate leaf of Chrysalidocarpus lutescens. Can J Bot 60:2976–2998
Deschamp PA, Cooke TJ (1984) Causal mechanisms of leaf dimorphism in the aquatic angiosperm Callitriche heterophylla. Am J Bot 71:319–329
Donnelly PM, Bonetta D, Tsukaya H, Dengler R, Dengler NG (1999) Cell cycling and cell enlargement in developing leaves of Arabidopsis. Dev Biol 215:407–419
Efroni I, Eshed Y, Lifschitz E (2010) Morphogenesis of simple and compound leaves: a critical review. Plant Cell 22:1019–1032
Feild T, Sage T, Czerniak C (2005) Hydathodal leaf teeth of Chloranthus japonicas (Chlorantheceae) prevent guttation-induced flooding of mesophyll. Plant Cell Environ 28:1179–1190
Fisher JB, Rutishauser R (1990) Leaves and epiphyllous shoots in Chisocheton (Meliaceae), a continuum of woody leaf and stem axes. Can J Bot 68:2316–2328
Fonseca CA, Overton JM, Collins B, Westoby M (2000) Shifts in trait-combinations along rainfall and phosphorus gradients. J Ecol 158:509–525
Fujikura U, Horiguchi G, Tsukaya H (2007) Dissection of enhanced cell expansion processes in leaves triggered by defect in cell proliferation, with reference to roles of endoreduplication. Plant Cell Physiol 48:278–286
Fujikura U, Horiguchi G, Ponce MR, Micol JL, Tsukaya H (2009) Coordination of cell proliferation and cell expansion mediated by ribosome-related processes in the leaves of Arabidopsis thaliana. Plant J 59:499–508
Fukuda T, Yokoyama J, Tsukaya H (2003) The evolutionary origin of indeterminate leaves in Meliaceae: phylogenetic relationships among species in the genera Chisocheton and Guarea, as inferred from sequences of chloroplast DNA. Int J Plant Sci 164:13–24
Fukushima K, Fujita H, Yamaguchi T, Masayoshi K, Tsukaya H, Hasebe M (2015) Oriented cell division shapes carnivorous pitcher leaves of Sarracenia purpurea. Nat Commun 6:6450. https://doi.org/10.1038/ncomms7450
Givnish TJ (1987) Comparative studies of leaf form: assessing the relative roles of selective pressures and phylogenetic constrains. New Phytol 106:131–160
Givnish TJ, Kriebel R (2017) Causes of ecological gradients in leaf margin entirety: evaluating the roles of biomechanics, hydraulics, vein geometry, and bud packing. Am J Bot 104:354–366
Gleissberg S, Groot EP, Schmalz M, Eichert M, Kölsch A, Hutter S (2015) Developmental events leading to peltate leaf structure in Tropaeolum majus (Tropaeolaceae) are associated with expression domain changes of a YABBY gene. Dev Genes Evol 215:313–319
Guo P, Yoshimura A, Ishikawa N, Yamaguchi T, Guo Y, Tsukaya H (2015) Comparative analysis of the RTFL peptide family on the control of plant organogenesis. J Plant Res 128:497–510
Gupta MD, Nath U (2015) Divergence in patterns of leaf growth polarity is associated with the expression divergence of miR396. Plant Cell 27:2785–2799
Ha CM, Kim GT, Kim BC, Jun JH, Soh MS, Ueno Y, Machida Y, Tsukaya H, Nam H-G (2003) The BLADE-ON PETIOLE gene controls leaf pattern formation through regulation of meristematic activity. Development 130:161–172
Harrison J, Möller M, Langdale J, Cronk Q, Hudson A (2005) The role of KNOX genes in the evolution of morphological novelty in Streptocarpus. Plant Cell 17:430–443
Hay A, Tsiantis M (2010) KNOX genes: versatile regulators of plant development and diversity. Development 137:3153–3165
Hay A, Kaur H, Phillips A, Hedden S, Hake S, Tsiantis M (2002) The gibberellin pathway mediates KNOTTED1-type homeobox function in plants with different body plans. Curr Biol 12:1557–1565
Hirayama Y, Yamada T, Oya Y, Ito M, Kato M, Imaichi R (2007) Expression patterns of class I KNOX and YABBY genes in Ruscus aculeatus (Asparagaceae) with implications for phylloclade homology. Dev Genes Evol 217:363–372
Hofer J, Turner L, Hellens R, Ambrose M, Matthews P, Michael A, Ellis N (1997) UNIFOLIATA regulates leaf and flower morphogenesis in pea. Curr Biol 7:581–587
Horiguchi G, Kim GT, Tsukaya H (2005) The transcription factor AtGRF5 and the transcription coactivator AN3 regulate cell proliferation in leaf primordia of Arabidopsis thaliana. Plant J 43:68–78
Horiguchi G, Nakayama H, Ishikawa N, Kubo M, Demura T, Fukuda H, Tsukaya H (2011) ANGUSTIFOLIA3 plays roles in adaxial/abaxial patterning and growth in leaf morphogenesis. Plant Cell Physiol 52:112–124
Ichihashi Y, Kawade K, Usami T, Horiguchi G, Takahashi T, Tsukaya H (2011) Key proliferative activity in the junction between the leaf blade and the leaf petiole of Arabidopsis thaliana. Plant Physiol 157:1151–1162
Ichihashi Y, Aquilar-Martinez JA, Farhi M, Chitwood DH, Kumar R, Milon LV, Peng J, Maloof JN, Sinha N (2014) Evolutionary developmental transcriptomics reveals a gene network module regulating interspecific diversity in plant leaf shape. Proc Natl Acad Sci U S A 111:E2616–E2621
Ikeuchi M, Igarashi H, Okada K, Tsukaya H (2014) Acropetal leaflet initiation of Eschscholzia californica is achieved by constant spacing of leaflets and differential growth of leaf. Planta 240:125–135
Imaichi R, Kato M (1992) Comparative leaf development of Osmunda lancea and Osmunda japonica (Osmundaceae) – heterochronic origin of rheophytic stenophylly. Bot Mag Tokyo 105:199–213
Ishikawa N, Takahashi H, Nakazono M, Tsukaya H (2017) Molecular bases for phyllomorph development in a one-leaf plant, Monophyllaea glabra. Am J Bot 104:233–240
Jasinski S, Piazza P, Craft J, Hay A, Woolley L, Rieu I, Phillips A, Hedden P, Tsiantis M (2005) KNOX action in Arabidopsis is mediated by coordinate regulation of cytokinin and gibberellin activities. Curr Biol 15:1560–1565
Jönsson H, Heisler MG, Shapiro BE, Meyerowitz EM, Mijolsness E (2006) An auxin-driven polarized transport model for phyllotaxis. Proc Natl Acad Sci U S A 103:1633–1638
Kane M, Albert LS (1987) Abscisic-acid induces aerial leaf morphology and vasculature in submerged Hippuris vulgaris L. Aquat Bot 28:81–88
Kato M, Imaichi R (1992a) Leaf anatomy of tropical fern rheophytes, with its evolutionary and ecological implications. Can J Bot 70:165–174
Kato M, Imaichi R (1992b) A broad-leaved variant of the fern rheophyte, Tectaria lobbii. Int J Plant Sci 153:212–216
Kawamura E, Horiguchi G, Tsukaya H (2010) Mechanisms of leaf tooth formation in Arabidopsis. Plant J 62:429–441
Kazama T, Ichihashi Y, Murata S, Tsukaya H (2010) The mechanism of cell cycle arrest front progression explained by a KLUH/CYP78A5-dependent mobile growth factor in developing leaves of Arabidopsis thaliana. Plant Cell Physiol 51:1046–1054
Kim JH, Kende H (2004) A transcriptional coactivator, AtGIF1, is involved in regulating leaf growth and morphology in Arabidopsis. Proc Natl Acad Sci U S A 101:13374–13379
Kim JH, Tsukaya H (2015) Regulation of plant growth and development by the GROWTH-REGULATING FACTOR and GRF-INTERACTING FACTOR duo. J Exp Bot 66:6093–6107. https://doi.org/10.1093/jxb/erv349
Kim G-T, Tsukaya H, Uchimiya H (1998) The ROTUNDIFOLIA3 gene of Arabidopsis thaliana encodes a new member of the cytochrome P450 family that is required for the regulated polar elongation of leaf cells. Genes Dev 12:2381–2391
Kim M, McCormick S, Timmermans M, Sinha N (2003) The expression domain of PHANTASTICA determines leaflet placement in compound leaves. Nature 424:438–443
Kim G-T, Yano S, Kozuka T, Tsukaya H (2005) Photomorphogenesis of leaves: shade-avoidance syndrome and differentiation of sun/shade leaves. Photochem Photobiol Sci 4:770–774
Kobayashi K, Baba S, Obayashi T, Sato M, Toyooka K, Karänen M, Aro EM, Fukaki H, Ohta H, Sugimoto K, Masuda T (2012) Regulation of root greening by light and auxin/cytokinin signaling in Arabidopsis. Plant Cell 24:1081–1095
Koenig D, Bayer E, Kang J, Kuhlemeier C, Sinha N (2009) Auxin patterns Solanum lycopersicum leaf morphogenesis. Development 136:2997–3006
Kozuka T, Kong SG, Nagatani A (2011) Tissue-autonomous promotion of palisade cell development by phototropin2 in Arabidopsis. Plant Cell 23:3684–3695
Kuwabara A, Ikegami K, Koshiba T, Nagata T (2003) Effects of ethylene and abscisic acid upon heterophylly in Ludwigia arcuata (Onagraceae). Planta 217:880–887
Lee YK, Kim G-T, Kim I-J, Park J, Kwak S-S, Choi G, Chung W-I (2006) LONGIFOLIA1 and LONGIFOLIA2, two homologous genes, regulate longitudinal cell elongation in Arabidopsis. Development 133:4305–4314
Leigh A, Close JD, Ball MC, Siebke K, Nicotra AB (2006) Leaf cooling curves: measuring leaf temperature in sunlight. Funct Plant Biol 33:515–519
Li Y, Zheng L, Corke F, Smith C, Bevan MW (2008) Control of final seed and organ size by the DA1 gene family in Arabidopsis thaliana. Genes Dev 2:1331–1336
Mano E, Horiguchi G, Tsukaya H (2006) Gravitropism in leaves of Arabidopsis thaliana (L.) Heynh. Plant Cell Physiol 47:217–223
Martorell C, Ezcurra E (2007) The narrow-leaf syndrome: a functional and evolutionary approach to the form of fog-harvesting rosette plants. Oecologia 151:561–573
Minamisawa N, Sato M, Cho K-H, Ueno H, Takeuchi K, Kajiwara M, Yamato KT, Ohyama K, Toyooka K, Kim G-T, Horiguchi G, Takano H, Ueda T, Tsukaya H (2011) AUNGUSTIFOLIA, a plant homolog of CtBP/BARS, functions outside the nucleus. Plant J 68:788–799
Nakata M, Matsumoto N, Tsugeki R, Rikirsch E, Laux T, Okada K (2012) Roles of the middle domain-specific WUSCHEL-RELATED HOMEOBOX genes in early development of leaves in Arabidopsis. Plant Cell 24:519–535
Nakayama H, Yamaguchi T, Tsukaya H (2010) Expression patterns of AaDL, a CRABS CLAW ortholog in Asparagus asparagoides (Asparagaceae), demonstrate a stepwise evolution of CRC/DL subfamily of YABBY genes. Am J Bot 97:591–600
Nakayama H, Yamaguchi T, Tsukaya H (2013) Modification and co-option of leaf developmental programs for the acquisition of flat structures in monocots: unifacial leaves in Juncus and cladodes in Asparagus. Front Plant Sci 4:248. https://doi.org/10.3389/fpls.2013.00248
Nakayama H, Nakayama N, Seiki S, Kojima M, Sakakibara H, Sinha N, Kimura S (2014) Regulation of the KNOX-GA gene module induces heterophyllic alteration in North American lake cress. Plant Cell 26:4733–4748
Nardmann J, Werr W (2013) Symplesiomorphies in the WUSCHEL clade suggest that the last common ancestor of seed plants contained at least four independent stem cell niches. New Phytol 199:1081–1092
Narita NN, Moore S, Horiguchi G, Kubo M, Demura T, Fukuda H, Goodrish J, Tsukaya H (2004) Overexpression of a novel small peptide ROTUNDIFOLIA4 decreases cell proliferation and alters leaf shape in Arabidopsis thaliana. Plant J 38:699–713
Nelissen H, Rymen B, Jilumaru Y, Demuynck K, Van Lijsebettens M, Kamiya Y, Inzé D, Beemster GTS (2012) A local maximum in gibberellin levels regulates maize leaf growth by spatial control of cell division. Curr Biol 22:1183–1187
Nelissen H, Eeckhout D, Demuynck K, Persiau G, Walton A, van bel M, Vervoot M, Candaele J, De Block J, Aesaert S, Van Lijsebettens M, Goormachtig S, Vandepoele K, Van Leene J, Muszynski M, Gevaert K, Inzé D, De Jaeger G (2015) Dynamic changes in ANGUSTIFOLIA3 complex composition reveal a growth regulatory mechanism in the maize leaf. Plant Cell 27:1605–1619
Nicotra AB, Cosgrove MJ, Cowling A, Schilichting CD, Jones CS (2008) Leaf shape linked to photosynthetic rates and temperature optima in south African Pelargonium species. Oecologia 154:625–635
Nicotra AB, Leigh A, Boyce CK, Jones CS, Niklas KJ, Royer DL, Tsukaya H (2011) The evolution and functional significance of leaf shape in the angiosperms. Funct Plant Biol 38:535–552
Nowak JS, Bolduc N, Dengler NG, Posluszny U (2011) Compound leaf development in the palm Chamaedorea elegans is KNOX-independent. Am J Bot 98:1575–1582
Ohnishi T, Szatmari A-M, Watanabe B, Fujita S, Bancos S, Koncz C, Lafos M, Shibata K, Yokota T, Sakata K, Yokota T, Sakata K, Szakeres M, Mizutani M (2006) C-23 hydroxylation by Arabidopsis CYP90C1 and CYP90D1 reveals a novel shortcut in Brassinosteroid biosynthesis. Plant Cell 18:3275–3288
Okajima Y, Taneda H, Noguchi K, Terashima I (2012) Optimum leaf size predicted by a novel leaf energy balance model incorporating dependencies of photosynthesis on light and temperature. Ecol Res 27:333–346
Omidbakhshfard MA, Proost S, Fujikura U, Mueller-Roeder B (2015) Growth-Regulating Factors (GRFs): a small transcription factor family with important functions in plant biology. Mol Plant 8:998–1010
Poethig RS (2013) Vegetative phase change and shoot maturation in plants. Curr Top Dev Biol 105:125–152
Royer DL, Wilf P, Janesko DA, Kowalski EA, Dilcher DL (2005) Correlations of climate and plant ecology to leaf size and shape: potential proxies for the fossil record. Am J Bot 92:1141–1151
Royer DL, Meyerson LA, Robertson KM, Adams JM (2009) Phenotypic plasticity of leaf shape along a temperature gradient in Acer rubrum. PLoS One 4:e7653
Sakamoto T, Kamiya N, Ueguchi-Tanaka M, Iwahori S, Matsuoka M (2001) KNOX homeodomain protein directly suppresses the expression of a gibberellin biosynthetic gene in the tobacco shoot apical meristem. Genes Dev 15:581–590
Schmerler SB, Clement WL, Beauliue JM, Chatelet DS, Sack L, Donoghue MJ, Edwards EJ (2012) Evolution of leaf form correlates with tropical-temperate transitions in Viburnum (Adoxaceae). Proc Roy Soc B 279:3905–3913
Smith RS, Guyomarc’h S, Mandel T, Reinhardt D, Kuhlemeier C, Prusinkiewicz P (2006) A plausible model of phyllotaxis. Proc Natl Acad Sci U S A 103:1301–1306
Steingraeber DA, Fisher JB (1986) Indeterminate growth of leaves in Guarea (Meliaceae): a twig analogue. Am J Bot 73:852–862
Sussex IM (1951) Experiments on the cause of dorsiventrality in leaves. Nature 167:651–652
Takahashi H, Iwakawa H, Ishibashi N, Kojima S, Matsumura Y, Prananingrum P, Iwasaki M, Takahashi A, Ikezaki M, Luo LL, Kobayashi T, Machida Y, Machida C (2013) Meta-analyses of microarrays of Arabidopsis asymmetric leaves1 (as1), as2 and their modifying mutants reveal a critical role for the ETT pathway in stabilization of adaxial-abaxial patterning and cell division during leaf development. Plant Cell Physiol 54:418–431
Tozer WC, Rice B, Westoby M (2015) Evolutionary convergence of leaf width and its correlates. Am J Bot 102:367–378
Tsuge T, Tsukaya H, Uchimiya H (1996) Two independent and polarized processes of cell elongation regulate leaf blade expansion in Arabidopsis thaliana (L.). Heynh. Develop 122:1589–1600
Tsukaya H (1997) Determination of the unequal fate of cotyledons of a one-leaf plant, Monophyllaea. Development 124:1275–1280
Tsukaya H (2000) The role of meristematic activities in the formation of leaf blades. J Plant Res 113:119–126
Tsukaya H (2002a) The leaf index: heteroblasty, natural variation, and the genetic control of polar processes of leaf expansion. Plant Cell Physiol 43:372–378
Tsukaya H (2002b) Leaf anatomy of a rheophyte, Dendranthema yoshinaganthum (Asteraceae), and of hybrids between D. yoshinaganthum and a closely related non-rheophyte species, D. indicum. J Plant Res 115:329–333
Tsukaya H (2006) Mechanism of leaf shape determination. Annu Rev Plant Biol 57:477–496
Tsukaya H (2013) Leaf development, 2nd edn. The Arabidopsis Book 11:e0163, American Society of Plant Biologists, Rockville. doi:https://doi.org/10.1199/tab.0072
Tsukaya H (2014) Comparative leaf development in angiosperms. Curr Opin Plant Biol 17:103–109
Tsukaya H (2018) How have leaves of mycoheterotrophic plants evolved—from the view point of a developmental biologist. New Phytol 217:1401–1406 https://doi.org/10.1111/nph.14994
Tsukaya H, Uchimiya H (1997) Genetic analyses of developmental control of serrated margin of leaf blades in Arabidopsis: combination of a mutational analysis of leaf morphogenesis with characterization of a specific marker gene expressed in hydathodes and stipules. Mol Gen Genet 256:231–238
Tsukaya H, Tsuge T, Uchimiya H (1994) The cotyledon: a superior system for studies of leaf development. Planta 195:309–312
Tsukaya H, Tsujino R, Ikeuchi M, Isshiki Y, Kono M, Takeuchi T, Araki T (2007) Morphological variation in leaf shape in Ainsliaea apiculata with special reference to the endemic characters of populations on Yakushima Island, Japan. J Plant Res 120:351–358
Usami T, Horiguchi G, Yano S, Tsukaya H (2009) The more and smaller cells mutants of Arabidopsis thaliana identify novel roles for SQUAMOSA PROMOTER BINDING PROTEIN-LIKE genes in the control of heteroblasty. Development 136:955–964
Usukura M, Imaichi R, Kato M (1994) Leaf morphology of a facultative rheophyte, Farfugium japonicum var. luchuense (Compositae). J Plant Res 107:263–267
van Steenis CGGJ (1981) Rheophytes of the world. Sijthoff & Noordhoff, Alphen aan den Rijn, The Netherlands
Vlad D, Kierzkowski D, Rast M, Vuolo F, Ioio RD, Galinha C, Gan X, Haijeidari M, Hay A, Smith RS, Huijser P, Bailey CD, Tsiantis M (2014) Leaf shape evolution through duplication, regulatory diversification, and loss of a homeobox gene. Science 343:780–783
Waites R, Hudson A (1995) Phantastica: a gene required for dorsoventrality of leaves in Antirrhinum majus. Development 121:2143–2154
Wang H, Jones B, Li Z, Frasse P, Delalande C, Regad F, Chaabouni S, Latché A, Pech J-C, Bouzayen M (2005) The tomato Aux/IAA transcription factor IAA9 is involved in fruit development and leaf morphogenesis. Plant Cell 17:2676–2692
Wang J-W, Park MY, Wang L-J, Koo YJ, Chen X-Y, Weigel D, Poethig RS (2011) MiRNA control of vegetative phase changes in trees. PLoS Genet 7:e1002012
Wen J, Lease KA, Walker JC (2004) DVL, a novel class of small polypeptides: overexpression alters Arabidopsis development. Plant J 37:668–677
Xu L, Xu Y, Dong A, Sun Y, Pi L, Xu Y, Huang H (2003) Novel as1 and as2 defects in leaf adaxial-abaxial polarity reveal the requirement for ASYMMETRIC LEAVES1 and 2 and ERECTA functions in specifying leaf adaxial identity. Development 130:4097–4107
Yamada T, Okuda T, Abdullah M, Awang M, Furukawa A (2000) The leaf development process and its significance for reducing self-shading of a traopical pioneer tree species. Oecologia 125:476–482
Yamaguchi T, Ikeuchi M, Tsukaya H (2013) Chapter 11: ROTUNDIFOLIA4. In: Matsuzaki Y (ed) Handbook of biologically active peptides, 2nd edn. Elsevier, San Diego, pp 53–57
Yamaguchi T, Nagasawa N, Kawasaki S, Matsuoka M, Nagato Y, Hirano H-Y (2004) The YABBY gene DROOPING LEAF regulates carpel specification and midrib development in Oryza sativa. Plant Cell 16:500–509
Yamaguchi T, Yano S, Tsukaya H (2010) Genetic framework for flattened leaf blade formation in unifacial leaves of Juncus prismatocarpus. Plant Cell 22:2141–2155
Yano S, Terashima I (2004) Developmental process of sun and shade leaves in Chenopodium album L. Plant Cell Physiol 27:781–793
Yorifuji E, Ishikawa N, Okada H, Tsukaya H (2015) Arundina graminifolia var. revoluta (Arethuseae, Orchidaceae) has fern-type rheophyte characteristics in the leaves. J Plant Res 128:239–247
Young JP, Dengler NG, Horton RF (1987) Heterophylly in Ranunculus flabellis: the effect of abscisic acid on leaf anatomy. Ann Bot 60:117–125
Zurakowski K, Gifford EM (1988) Quantitative studies of pinnule development in the ferns Adiantum raddianum and Cheilanthes viridis. Am J Bot 75:1559–1570
Acknowledgments
This work was supported by the Bio-Next project in NINS, the Japan Society for the Promotion of Science (Grants-in-Aid for Creative Scientific Research and Scientific Research A), The Ministry of Education, Culture, Sports, Science and Technology, Japan (Scientific Research on Priority Areas and Scientific Research on Innovative Areas No. 25113002). In addition, the author thanks reviewers of the manuscript who helped him to polish it. Dr. Hiroyuki Koga of the University of Tokyo also kindly checked the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Tsukaya, H. (2018). A Consideration of Leaf Shape Evolution in the Context of the Primary Function of the Leaf as a Photosynthetic Organ. In: Adams III, W., Terashima, I. (eds) The Leaf: A Platform for Performing Photosynthesis. Advances in Photosynthesis and Respiration, vol 44. Springer, Cham. https://doi.org/10.1007/978-3-319-93594-2_1
Download citation
DOI: https://doi.org/10.1007/978-3-319-93594-2_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-93592-8
Online ISBN: 978-3-319-93594-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)