Abstract
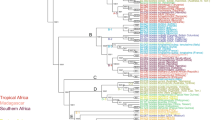
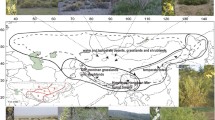
Castela Turpin is composed of 16 known species and is found throughout edaphically dry zones across the American continents from the Sonoran Desert to the southern South American Chaco biogeographic region and Caribbean seasonally dry tropical forests (SDTF). Castela is most diverse in the SDTF of the Greater Antilles, with seven of the 16 known species occurring there. We reconstructed the phylogeny of Castela sampling 15 of the 16 known species using a novel plastome dataset. Our phylogeny was used to test the biogeographic history and evolution of morphological characters across the clade. Castela was composed of four major subclades: the Holacantha, Depressa, Caribbean, and Chaco clades. Castela most likely originated in the Mojave/Sonoran Desert/Baja California region and then moved into the Chihuahuan Desert, Chaco, Greater and Lesser Antilles and northern South America, suggesting multiple long-distance dispersal events. Taxa occurring in arid areas, such as the Sonoran and Chihuahuan deserts and parts of the Caribbean SDTF, have a tendency to quickly lose or drastically reduce the size of their leaves, and most photosynthesis appears to be taken over by photosynthetic stems. The Holacantha clade exhibits the plesiomorphic yellow-green flowers so common in much of the rest of the family, while Castela s.s. mostly has derived red/rose-colored flowers. The putative Miocene origin of Castela and subsequent diversification is likely tied to the expansion of arid lands in the Americas; however, speciation within those clades may have been driven by peripheral isolate speciation and vicariance through habitat fragmentation resulting from more recent climatic change.




Similar content being viewed by others
References
Bell DS, Herskovits T (2013) A newly discovered large and significant population of Castela emoyri (Emoryi’s Crucifix Thorn, Simaroubaceae) in California. Aliso 31:43–47
Borhidi AL (1991) Evolutionary centres and migratory routes of the flora of limestone cliffs: In Phytogeography and Vegetation Ecology of Cuba. Akadémiai Kiadó, Budapest. pp. 275–279
Borhidi AL (1996) Phytogeography and vegetation ecology of Cuba. Akadémiai Kiadó, Budapest
Cabrera AL, Willink A (1973) Biogeografía de América Latina. Programa Regional de Desarrollo Científico y Tecnológico, Departamento de Asuntos Científicos, Secretaria General de la Organización de los Estados Americanos, Washington, District of Columbia
Clayton JW (2011) Simaroubaceae. In: Kubitzki K (ed) The families and genera of vascular plants. Vol. X. Flowering plants. Eudicots: Sapindales, Cucurbitales, Myrtaceae. Berlin, Springer, pp 408–423
Clayton JW, Fernando ES, Soltis PS, Soltis DS (2007) Molecular phylogeny of the tree-of-heaven family (Simaroubaceae) based on chloroplast and nuclear markers. Int J Pl Sci 168:1325–1339
Clayton JW, Soltis PS, Soltis DS (2009) Recent long-distance dispersal overshadows ancient biogeographical patterns in a pantropical angiosperm family (Simaroubaceae, Sapindales). Syst Biol 58:395–410
Crayn DM, Fernando ES, Gadek PA, Quinn CJ (1995) A reassessment of the familial affinity of the Mexican genus Recchia Moçiño & Sessé ex DC. Brittonia 47:397–402
Cronquist A (1944a) Studies in the Simaroubaceae, 1: The genus Castela. J Arnold Arb 25:122–128
Cronquist A (1944b) Studies in the Simaroubaceae–IV. Resume of the American Genera Brittonia 5:128–147
Cronquist A (1945) Additional notes on the Simaroubaceae. Brittonia 5:469–470
Doyle JJ, Doyle JL (1989) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phyto Bull 19:11–15
Durham JW (1985) Movement of the Caribbean plate and its importance for biogeography in the Caribbean. Geology 13:123–125
Engler A (1931) Simaroubaceae. Die Natürlichen Pflanzenfamilien, Band 19a:359–405
Engler HGA, Prantl KAU (1896) Die Natürlichen Pflanzenfamilien nebst ihren Gattungen und wichtigeren Arten, insbesondere den Nutzpflanzen, unter Mitwirkung zahlreicher hervorragender Fachgelehrten begründet. 4:218–220
Fernando ES, Gadek PA, Quinn CJ (1995) Simaroubaceae, an artificial construct: evidence from rbcL sequence variation. Am J Bot 82:92–103
Frey JK (1993) Modes of peripheral isolate formation and speciation. Syst Biol 42:373–381
GBIF.org (13 April 2021) GBIF Occurrence Download https://doi.org/10.15468/dl.y82yef
Harris AJ, Ickert-Bond S, Rodríguez A (2018) Long distance dispersal in the assembly of floras: a review of progress and prospects in North America. J Syst Evol 56:430–448
Judd WS, Campbell CS, Kellogg EA, Stevens PF, Donoghue MJ (2016) Plant systematics: a phylogenetic approach, 4th edn. Sinauer Associates Inc, Sunderland, Massachusettes
Katoh K, Standley DM (2016) A simple method to control over-alignment in the MAFFT multiple sequence alignment program. Bioinformatics 32:1933–1942
Liogier AH (1985) La Flora de la Española III. Universidad Central del Este, R.D, San Pedro de Macorís, Dominica
Logacheva MD, Shipunov AB (2017) Phylogenomic analysis of Picramnia, Alvaradoa, and Leitneria supports the independent Picramniales. J Syst Evol 55:171–176
Maddison WP, Maddison DR (2017) Mesquite: a modular system for evolutionary analysis. Version 3.2. http://mesquiteproject.org
Majure LC, Puente R, Griffith MP, Judd WS, Soltis PS, Soltis DS (2012) Phylogeny of Opuntia s.s. (Cactaceae): clade delineation, geographic origins, and reticulate evolution. Am J Bot 99:847–864
Majure LC, Baker M, Cloud-Hughes M, Salywon A, Neubig KM (2019) Phylogenomics in Cactaceae: a case study using the chollas sensu lato (Cylindropuntieae, Opuntioideae) reveals a common pattern out of the Chihuahuan/Sonoran desert. Am J Bot 106:1–19
Majure LC, Clase T, Blankenship A, Noa-Monzón A (2021a) A new species of Picrasma, P. nanophylla (Simaroubaceae), from the Dominican Republic. Brittonia https://doi.org/10.1007/s12228-021-09656-x
Majure LC, Barrios D, Díaz E, Zumwalde B, Testo W, Negrón-Ortiz V (2021b) Pleistocene drying underlies the evolutionary history of the Caribbean endemic, insular giant, Consolea (Opuntioideae). Am J Bot 108:1–16. https://doi.org/10.1002/ajb2.1610
Majure LC, Blankenship A, Grinage A, Noa-Monzón A (2021c) Castela (Simaroubaceae), an impressive New World radiation of thorny shrubs destined for edaphically dry habitats. (dataset on FigShare)
Majure LC, Pham K, Clase T (in press) Castela senticosa (Simaroubaceae: Sapindales), a new species from the Caribbean clade endemic to seasonally dry tropical forest on Hispaniola. Syst Bot
Martínez-Berdeja A, Ezcurra E, Sanders AC (2015) Delayed seed dispersal in California desert. Madroño 62:21–32
Medeiros AS, Drezner TD (2012) Vegetation, climate, and soil relationships across the Sonoran Desert. Ecoscience 19:1–13
Moran R, Felger R (1968) Castela polyandra, a new species in a new section; union of Holacantha with Castela (Simaroubaceae). Trans San Diego Soc Nat Hist 15:33–40
Noa-Monzón A, González-Guitiérrez PA (2019) Picrasma pauciflora (Simaroubaceae), a new species of from the NE coast of Cuba. Willdenowia 49:187–191
Pennington T, Lavin M, Prado DE, Pendry CE, Pell SK, Butterworth CA (2004) Historical climate change and speciation: neotropical seasonally dry forest plants show patterns of both Tertiary and Quaternary diversification. Phil Trans Royal Soc London B 359:515–537
Persson C (2001) Phylogenetic relationships in Polygalaceae based on plastid DNA sequences from the trnL-F region. Taxon 50:763–779
Pirani JR (1987) Simaroubaceae. In: Spichiger R (ed) Flora de Paraguay. Editions des Conservatoire et Jardin botaniques de la Ville de Genève. pp 1–28
Pirani JR, Majure LC, Devecchi MF (2021) An updated account of Simaroubaceae with an emphasis on American taxa. Braz J Bot (this issue)
Powell AM Worthington RD (2018) Flowering plants of the Trans-Pecos Texas and adjacent areas. Sida, Bot. Misc. 49. Botanical Research Institute of Texas, Fort Worth, Texas
Prado DE (1993) What is the Gran Chaco vegetation in South America? I. A review. Contribution to the study of flora and vegetation of the Chaco. V. Candollea 48:145–172
Ree RH, Smith SA (2008) Maximum likelihood inference of geographic range evolution by dispersal, local extinction and cladogenesis. Syst Biol 57:4–14
Riddle BR, Hafner DJ (2006) A step-wise approach to integrating phylogeographic and phylogenetic biogeographic perspectives on the history of a core North American warm deserts biota. J Arid Environ 66:435–461
Ripma LA, Simpson MG, Hasenstab-Lehman K (2014) Geneious! Simplified genome skimming methods for phylogenetic systematic studies: a case study in Oreocarya (Boraginaceae). Appl Plant Sci. https://doi.org/10.3732/apps.1400062
Saina JK, Li ZZ, Gichira AW, Liao YY (2018) The complete chloroplast genome sequence of tree of heaven (Ailanthus altissimus (Mill.)) (Sapindales: Simaroubaceae), an important pantropical tree. Int J Mol Sci 19:929. doi: https://doi.org/10.3390/ijms19040929
Simpson MG, Johnson LA, Villaverde T, Guilliams CM (2017) American amphitropical disjuncts: perspectives from vascular plant analyses and prospects for future research. Am J Bot 104:1600–1650
Solbrig OT (1972) The floristic disjunctions between the “Monte” of Argentina and the “Sonoran Desert” in Mexico and the United States. Ann Mo Bot Gard 59:218–223
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30:1312–1313
Straub SCK, Parks M, Weitemier K, Fishbein M, Cronn RC, Liston A (2012) Navigating the tip of the genomic iceberg: next-generation sequencing for plant systematics. Am J Bot 99:349–364
Steadman DS, Franklin J (2015) Changes in a West Indian bird community since the last Pleistocene. J Biogeogr 42:426–438
Thiers B (2017) Index Herbariorum: a global directory of public herbaria and associated staff. New York Botanical Garden's Virtual Herbarium. http://sweetgum.nybg.org/scien ce/ih/
Thomas WW (1990) The American genera of Simaroubaceae and their distribution. Acta Bot Bras 4:11–18
Thomas WW, Mitchell JD, Pell S, Noa Monzón A (2011) Euleria (Anacardiaceae) is Picrasma (Simaroubaceae): the genus Picrasma in Cuba. Brittonia 63:419–424
Turpin P (1806) Castela: polygamie monoecie Lin Ann Mus D’histoire Nat 7:79–81
Wilson JS, Pitts JP (2010) Illuminating the lack of consensus among descriptions of earth history data in the North American deserts: a resource for biologists. Progr Phys Geogr 34:419–441
Yu Y, Harris AJ, He XJ (2010) S-DIVA (statistical dispersal-vicariance analysis): a tool for inferring biogeographic histories. Mol Phylogenet Evol 56:848–850
Yu Y, Harris AJ, Blair C, He XJ (2015) RASP (Reconstruct Ancestral State in Phylogenies): a tool for historical biogeography. Mol Phylogenet Evol 87:46–49
Acknowledgements
We would like to thank El Ministerio del Medio Ambiente for collecting permits in the Dominican Republic. Teodoro Clase (JBSD), Y. Encarnación (FLAS), W. Testo, and M. Bonifacino aided with fieldwork. This work was supported in part from start-up funds to L.C. Majure from the Florida Museum of Natural History and University of Florida, the National Science Foundation (GSS-1461496 to J. Franklin), and the Florida Museum of Natural History Biodiversity Fund to D. Steadman. We thank the following herbaria and their staff for specimen loans used for this work: ASU (E. Makings), NY (M. Pace), SRSU (A.M. Powell), US (M. Toner). J.R. Pirani and another anonymous reviewer provided helpful comments on an earlier version of this manuscript.
Author information
Authors and Affiliations
Contributions
LCM designed the study, acquired material for analysis, carried out laboratory work and data analysis, and wrote the paper. AB and AG carried out laboratory work, helped with analyses, and helped write the paper. AN helped write the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix 1
Appendix 1
Accessions used in our plastome dataset analyses with associated collector name and numbers, herbarium repositories (acronyms follow Thiers 2017 +), and their GenBank accession numbers. Accession numbers for data downloaded from GenBank are given in parentheses after the taxon name.
Simaroubaceae — Ailanthus altissimus (MG799542), Castela calcicola Morton 10,242 (US; SAMN17814252), Castela calcicola Acevedo 6521 (US; SAMN17814251), Castela coccinea Krapovikas 44,166 (ASU; SAMN20491061), Castela coccinea Piesko 8 (ASU; SAMN17814253), Castela depressa Jímenez 453 (US; SAMN17814254), Castela emoryi Baker 20,325 (FLAS; SAMN20491062), Castela emoryi Majure 5932 (DES, FLAS; SAMN17814255), Castela erecta Arnoldo 1638 (US; SAMN20491063), Castela erecta Acevedo 5315 (US; SAMN17814256), Castela galapageia Bowman s.n. (US; SAMN20491064), Castela jaquiniifolia Clemente 4301 (US; SAMN20491065), Castela macrophylla Britton 1843 (US; SAMN17814257), Castela peninsularis Rebman 2793 (ASU; SAMN20491066), Castela peninsularis Wiggins 16,151 (US; SAMN20491067), Castela polyandra Moran 11,895 (US; SAMN20491068), Castela polyandra Pinkava 14,228 (ASU; SAMN20491069), Castela sp. nov. Leonard 13,218 (US; SAMN17814258), Castela sp. nov. Majure 6629 (FLAS; SAMN17814259), Castela spinosa Roig 3931 (NY; SAMN20491071), Castela spinosa León 7219 (US; SAMN20491070), Castela stewartii Powell 6350 (SRSU; SAMN20491074), Castela stewartii Warnock 18,723 (SRSU; SAMN20491075), Castela stewartii Fenstermacher 1900 (SRSU; SAMN20491072), Castela stewartii Johnston 11,391 (ASU; SAMN20491073), Castela texana Saunders 1068 (US; SAMN20491076), Castela tweediei Pedersen 5125 (ASU; SAMN17814260), Castela tweediei DelPuerto 5372 (US; SAMN20491078), Castela tweediei Bonifacino 6956 (FLAS; SAMN20491077), Castela tweediei Hatschbach 72,435 (ASU; SAMN20491080), Castela tweediei Hatschbach 19,288 (NY; SAMN20491079), Eurycoma longifolia (MH751519), Leitneria floridana Judd 7022 (FLAS; SAMN17814261), Picrasma crenata Jardim 2795 (NY; SAMN20491081), Picrasma excelsa Trejo-Torres 2311 (FLAS; SAMN17814262), Picrasma mexicana Acevedo 375 (NY; SAMN20491082), Picrasma nanophylla Majure 6473 (FLAS; SAMN17814263), Picrasma nanophylla Clase 8980 (FLAS; SAMN20491083), Picrasma selleana Judd 4400 (FLAS; SAMN17814264), Picrasma tetramera Roig 16,788 (FLAS; SAMN20491084), Simarouba berteroana Majure 7811 (FLAS; SAMN20491085), Simarouba glauca Morris 4013 (FLAS; SAMN17814265). Rutaceae — Tetradium glabrifolium Tang 611 (FLAS; SAMN20520266), Zanthoxylum azuense Clase 8505 (JBSD; SAMN20520269), Zanthoxylum americanum Godfrey 79,818 (FLAS; SAMN20520268), Zanthoxylum alatum Péng 2803 (FLAS; SAMN20520267), Zanthoxylum flavum Majure 6280 (FLAS; SAMN17814268). Meliaceae — Azadirachta indica (NC023792), Melia azedarach (NC050650), Swietenia macrophylla (MH348156), Swietenia mahogani Majure 7108 (FLAS; SAMN17814266). Sapindaceae — Aesculus chinensis (MK648235), Koelreuteria paniculata (KY859413), Litchi chinensis (NC035238), Thouinia trifoliata Majure 6596 (FLAS, JBSD; SAMN17814267). Burseraceae — Commiphora foliacea (NC041103), Commiphora gileadensis (NC041104). Anacardiaceae — Anacardium occidentale (NC035235), Mangifera indica (NC035239), Pistacia chinensis (NC046786), Rhus potaninii (MT230556), Spondias mombin (NC035973).
Rights and permissions
About this article
Cite this article
Majure, L.C., Blankenship, A., Grinage, A. et al. Castela (Simaroubaceae), an impressive New World radiation of thorny shrubs destined for edaphically dry habitats. Braz. J. Bot 45, 237–249 (2022). https://doi.org/10.1007/s40415-021-00742-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-021-00742-8