Abstract
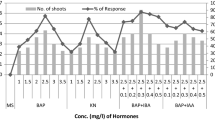
An important, endemic, and imperilled tropical medicinal plant called Tabernaemontana alternifolia is successfully micropropagated in this article. On Murashige and Skoog’s media that had thidiazuron (TDZ; 2.5, 5.0, 7.5, and 10 µM) supplied, high-frequency indirect shoot regeneration was induced from the callus produced from internode explants. After 12 weeks of culture, 10 µM TDZ produced the greatest number of shoots among the different TDZ concentrations examined (15.33 shoots). From the original callus, up to three subcultures could regenerate shoots. On full and half-strength MS media containing indole-3-butyric acid (IBA) after 6 weeks of culture, rooting of the shoots was seen. Rooting of shoots was facilitated by a half-strength MS medium supplemented with 5 µM IBA. After successfully being transplanted into a potting mixture including vermiculite and perlite and being raised there for 4 weeks, the plants were moved to one containing soil, sand, and farmyard manure and kept in the greenhouse. Inter simple sequence repeat (ISSR) and directed amplification of minisatellite DNA (DAMD) primer assays were used to assess the genetic stability of Tabernaemontana alternifolia micropropagated plants. When randomly chosen regenerated plants were compared to donor plants, a homogenous amplification profile was found, supporting the clonal fidelity of micropropagated plants. Morphometric analysis of regenerated plants was comparable with that of mother plants suggesting the genetic fidelity of regenerated plants.


Similar content being viewed by others
References
Abraham A, Cheruvathur MK, Mani B, Thomas TD (2010) A rapid in vitro multiplication system for commercial propagation of pharmaceutically important Cyclea peltata (Lam) Hook & Thoms. based on enhanced axillary branching. Ind Crop Prod 31:92–98
Ahn JH, Lee SY (2021) High-frequency mirotuberization by one-step culture without medium renewal from leaf explants of Pinellia ternata, a medicinal plant. Hotic Sci Technol 39:497–506
Banerjee S, Haider F, Bagchi GD, Samad A (2010) Regeneration of phytoplasma-free Artemisia roxburghiana Besser var. purpurascens (Jacq.) Hook plants using apical meristem. Plant Cell Tiss Org Cult 103:189–196
Bao MF, Yan JM, Cheng GG, Li XY, Liu YP, Li Y, Cai XH, Luo XX (2013) Cytotoxic indole alkaloids from Tabernaemontana divaricata. J Nat Prod 76:1406–1412
Dandin VS, Murthy HN (2012) Enhanced in vitro multiplication of Nothapodytes nimmoniana Graham using semisolid and liquid cultures and estimation of camptothecin in the regenerated plant. Acta Physiol Plant 24:1381–1386
Das A, Mooquammel Haque SK, Ghosh B, Nandagopal K, Jha TB (2015) Morphological and genetic characterization of field grown plants of Aloe vera L. Plant Tissue Cult Biotechnol 25:231–246
Gunasekera SP, Cordell G, Farnsworth NR (1980) Anticancer indole alkaloids of Ervatamia alternifolia. Phytochemistry 19:1213–1218
Hazarika BN (2003) Acclimatization of tissue-cultured plants. Curr Sci 85:1704–1712
Hiregoudar LV, Ashok Kumar HG, Murthy HN (2005) In vitro culture of Feronia limonia (L.) Swingle from hypocotyls and intermodal explants. Biol Plant 49:41–45
Huetteman CA, Preece JE (1993) Thidiazuron: a potent cytokinin for woody plant tissue culture. Plant Cell Tissue Org Cult 33:105–119
Ingkaninan K, Ijzerman AP, Taesotikult T, Verpoorte R (1999) Isolation of opioid-active compounds from Tabernaemontana pachysiphon leaves. J Pharm Pharmacol 51:1441–1446
Joshi P, Dhawan V (2007) Assessment of genetic fidelity of micropropagated Swertia chirayita plantlets by ISSR marker assay. Biol Plant 51:22–26
Kadapatti SS, Murthy HN (2021a) In vitro micropropagation of Andrographis macrobotrys. J Herbs Spices Med Plants 28:89–98
Kadapatti SS, Murthy HN (2021b) Rapid plant regeneration, analysis of genetic fidelity, and neoandrographolide in micropropagated plants of Andrographis alata (Vahl) Nees. J Genet Eng Biotechnol 19:20
Jirakiattikul Y, Rithichai P, Kwanthong P, Itharat A (2021) Effect of jasmonic acid elicitation period on enhancement of bioactive compounds and antioxidant activity in callus cultures of Hibiscus sabdariffa Linn. Hortic Environ Biotechnol 62:629–636
Lee HJ, Kim WS (2022) Effect of light and temperature on the long-term storage of in vitro Gerbera plantlets. Hotic Sci Technol 40:703–711
Leeuwenberg AJM (1994) A revision of Tabernaemontana two. The new world species and Stemmadenia (Series of revisions of Apocynaceae; No. 36) Royal Botanic Gardens, pp 450
Liu CZ, Murch SJ, El-Demerdash M, Saxena PK (2003) Regeneration of egyptian medicinal plant Artemisia judaica L. Plant Cell Rep 21:525–530
Meyer WE, Coppola JA, Goldman L (1973) Alkaloid studies VIII: isolation and characterization of alkaloids of Tabernaemontana alternifolia Wall and antifertility properties of coronarideine. J Pharm Sci 62:1799–1201
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:437–497
Murry MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic acid Res 8:4321–4325
Murthy BNS, Murch SJ, Saxena PK (1998) Thidiazuron: a potent regulator of in vitro plant morphogenesis. In Vitro Cell Dev Biol Plant 4:267–275
Naidoo CM, Naidoo Y, Dewir YH, Murthy HN, El-Hendawy S, Al-Suhaibani N (2021) Major bioactive alkaloids and biological activities of Tabernaemontana species (Apocynaceae). Plants 10:313
Negi D, Saxena S (2010) Ascertaining clonal fidelity of tissue culture raised plants of Bambusa balcooa Roxb. Using inter simple sequence repeat markers. New For 40:1–8
Phulwaria M, Rai MK, Patel AK, Kataria V, Shekhawat NS (2013) A genetically stable rooting protocol for propagating a threatened medicinal plant-Celastrus paniculatus. AoB Plants 5. https://doi.org/10.1093/aobpla/pls054
Rani V, Raina SN (2000) Genetic fidelity of organized meristem derived micropropagated plants: a critical appraisal. In Vitro Cell Dev Biol Plant 36:319–330
Ren X, Liu Y, Jeong BR (2020) Callus induction and browning suppression in tree peony Paeonia ostii ‘Fengdan’. Hortic Environ Biotechnol 61:591–600
Riahi L, Chograni H, Rejab FB, Romdhane MB, Masmoudi AS, Cherif A (2022) Efficient in vitro regeneration of the endangered species of Artemisia arborescens L. through direct organogenesis and impact on secondary metabolite production. Hortic Environ Biotechnol 63:439–450
Satishkumar T, Baskar R (2014) Renoprotective effect of Tabernaemontana alternifolia wall. leaves against paracetamol-induced renotoxicity in rats and detection of polyphenols by high-performance liquid chromatography-diode array detector-mass spectrometry analysis. J Acute Med 4:57–64
Scaramuzzi F, Apollonio G, D’Emerico S (2000) In vitro propagation of two species of commelinaceae from bud cultures and repeated subcultures on a growth regulator-free medium. In Vitro Cell Dev Biol Plant 36:379–382
Shen X, Castle WS, Gimitter FG (2010) In vitro shoot proliferation and root induction of the shoot tip explants from mature male plants of Casuarina cunninghamiana Miq. Hort Sci 45:797–800
Shinde S, Katewal PR, Shanbhag DD, Joseph KS, Murthy HN (2017) In vitro propagation of Artemisia japonica. J Herbs Spices Med Plants 23:36–43
Shinde S, Sebastain KS, Jain JR, Hanamathagouda MS, Murthy HN (2016) Efficient in vitro propagation of Artemisia nilagirica var. nilagirica (Indian wormwood) and assessment of genetic fidelity of micropropagated plants. Physiol Mol Biol Plants 22:595–603
Simoes AO, Endress ME, Conti E (2010) Systematics and character evolution of Tabernaemontaneae (Apocynaceae, Rauvolfioideae) based on molecular and morphological evidence. Taxon 59:772–790
Srivastava S, Singh MM, Kulshreshtha DK (2001) A new alkaloid and other anti-implantation principles from Tabernaemontana alternifolia. Planta Med 67:577–579
Van Beek T, Verpoorte R, Svendsen A, Leeuwenberg A, Bisset N (1984) Tabernaemontana L. (Apocynaceae): A review of its taxonomy, phytochemistry, ethnobotany and pharmacology. J Ethnopharmacol 10:1-156
World Conservation Monitoring Centre (1998) Tabernaemontana alternifolia. The IUCN Red List of Threatened Species 1998: e.T 35327A9927070 https://doi.org/10.2305/IUCN.UK.1998.RLTS.T35327A9927070.en
Acknowledgements
One (HNM) of the authors is thankful for the ‘Brain Pool (BP)’ program Grant No. 2022H1D3A2A02056665.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Compliance with ethical standards
Authors declare that they have no conflict of interest.
Additional information
Communicated by Jeong Hee Choi.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hosakatte Niranjana Murthy is co-corresponding author.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shinde, S., Jain, J.R., Kadapatti, S. et al. Micropropagation system and their genetic fidelity evaluation from regenerated plants by ISSR and DAMD markers of Tabernaemontana alternifolia L., an endangered medicinal woody species. Hortic. Environ. Biotechnol. 64, 849–857 (2023). https://doi.org/10.1007/s13580-023-00520-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-023-00520-3