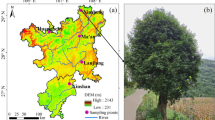
Abstract
Carpinus tschonoskii Maxim. exhibits rich leaf phenotypic variation and various leaf shapes, but few studies show why leaf phenotypic traits have such a large variation. Basic morphological markers may provide guidance for studying plant genetic variation and species protection and utilization. To study leaf phenotypic variations and the relationship between variation characteristics and climatic and geographical factors, phenotypic traits among natural populations were investigated. Results revealed that leaf phenotypes varied significantly among and within populations. Some populations had higher phenotypic diversity, while others had lower phenotypic diversity. Among the phenotypic traits, leaf area and petiole length had the most variation. Leaf index and primary lateral veins were the most stable phenotypes, which may be important reference indexes for phenotype identification in field investigations. There was a strong consistency between leaf phenotypic traits and geographical location. Plants in high latitudes tend to have longer leaves, and plants in low temperatures tend to have longer leaves and larger leaf perimeter. In addition, plants in areas with less rainfall have longer petioles. The 13 populations of C. tschonoskii can be divided into four branches by cluster analysis, and the results show a good relationship with the geographical location of each population. Additionally, some populations geographically isolated also had unique leaf phenotypes.




Similar content being viewed by others
References
Ackerly DD, Knight CA, Weiss SB, Barton K, Starmer KP (2002) Leaf size, specific leaf area and microhabitat distribution of chaparral woody plants: contrasting patterns in species level and community level analyses. Oecologia 130:449–457. https://doi.org/10.1007/s004420100805
Ali K (2018) Phenotypic characterization of Elaeagnus angustifolia using multivariate analysis. Ind Crops Prod 120:155–161. https://doi.org/10.1016/j.indcrop.2018.04.050
Barros FDV, Goulart MF, Telles SBS, Lovato MB, Valladares F, Lemos-Filho JPD (2012) Phenotypic plasticity to light of two congeneric trees from contrasting habitats: Brazilian Atlantic Forest versus cerrado (savanna). Plant Biol 14:208–215. https://doi.org/10.1111/j.1438-8677.2011.00474.x
Bi DZ, Chen D, Khayatnezhad M, Hashjin ZS, Li ZF, Ma YX (2021) Molecular identification and genetic diversity in Hypericum L.: a high value medicinal plant using RAPD markers. Genetika 53:393–405. https://doi.org/10.2298/GENSR2101393B
Chen R (1937) Illustrated manual of Chinese trees and shrubs. Chinese Agricultural Society, Beijing, p 166 (in Chinese)
Chen L (2016) Molecular markers and the application in genetic diversity research. In: Ding YL, Fang YM (eds) Special Topics in Plant Biology. China Forestry Publishing House, Beijing, pp 296–307 (in Chinese)
Clausen J, Keck DD, Hiesey WM (1940) Experimental studies on the nature of species. I. Effect of varied environments on western North American plants. Carnegie Institution of Washington Publication No. 520. Washington, DC
De Kort H, Prunier JG, Ducatez S, Honnay O, Baguette M, Stevens VM, Blanchet S (2021) Life history, climate and biogeography interactively affect worldwide genetic diversity of plant and animal populations. Nat Commun 12:516. https://doi.org/10.1038/s41467-021-20958-2
DeWoody JA, Harder AM, Mathur S, Willoughby JR (2021) The long-standing significance of genetic diversity in conservation. Mol Ecol 30:4147–4154. https://doi.org/10.1111/mec.16051
Fick SE, Hijmans RJ (2017) WorldClim 2: New 1-km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Frankham R (2005) Genetics and extinction. Biol Conserv 126:131–140. https://doi.org/10.1016/j.biocon.2005.05.002
García D, Zamora R, Gómez JM, Jordano P, Hódar JA (2000) Geographical variation in seed production, predation and abortion in Juniperus communis throughout its range in Europe. J Ecol 88:435–446. https://doi.org/10.1046/j.1365-2745.2000.00459.x
Glennon K, Cron G (2015) Climate and leaf shape relationships in four Helichrysum species from the Eastern Mountain Region of South Africa. Evol Ecol 29:657–678. https://doi.org/10.1007/s10682-015-9782-7
Gong YB, Yang M, Vamosi JC, Yang HM, Mu WX, Li JK, Wan T (2016) Wind or insect pollination? Ambophily in a subtropical gymnosperm Gnetum parvifolium (Gnetales). Plant Spec Biol 31:272–279. https://doi.org/10.1111/1442-1984.12112
Han S, Kang G, Park D, Kang H, Yoo B, Yoo E (2011) Solvent extracts of Carpinus tschonoskii suppress the expression of atopic inflammatory cytokines and chemokines in RAW264.7 macrophages and HaCaT keratinocytes. Planta Med 77:1431. https://doi.org/10.1055/s-0031-1282884
Jenczewski E, Prosperi JM, Ronfort J (1999) Evidence for gene flow between wild and cultivated Medicago sativa (Leguminosae) based on allozyme markers and quantitative traits. Am J Bot 86:677–687. https://doi.org/10.2307/2656577
Kang GJ, Kang NJ, Han SC, Koo DH, Kang HK, Yoo BS, Yoo ES (2012) The Chloroform Fraction of Carpinus tschonoskii Leaves Inhibits the Production of Inflammatory Mediators in HaCaT Keratinocytes and RAW264.7 Macrophages. Toxicol Res 28:255–262. https://doi.org/10.5487/TR.2012.28.4.255
Kim MK, Kim SC, Kang JI, Boo HJ, Hyun JW, Koh YS, Park DB, Yoo ES, Kang JH, Kang HK (2010) Neuroprotective effects of Carpinus tschonoskii MAX on 6-Hydroxydopamine-induced death of PC 12 cells. Biomol Ther 18:454–462. https://doi.org/10.4062/biomolther.2010.18.4.454
Koksheeva I, Kislov D, Tvorogov S, Doudkin R (2017) Relationships between leaf shape and climate in Rhododendron mucronulatum. Nord J Bot 35:618–626. https://doi.org/10.1111/njb.01486
Lande R (1988) Genetics and demography in biological conservation. Science 241:1455–1460. https://doi.org/10.1126/science.3420403
Li XX (2016) Genetic structure of rare and endangered woody plants. In: Ding YL (ed) Fang YM. China Forestry Publishing House, China, Special Topics in Plant Biology. Beijing, pp 296–307 (in Chinese)
Li PQ, Skvortsov AK (1999) Flora of China 4. Science Press, Beijing, pp 289–300
Li PQ, Zheng SX (1979) Flora Republicae Popularis Sinica 21. Science Press, China, Beijing, pp 84–85 (in Chinese)
Li M, Han HR, Kang FF (2005) Morphologic variation of leaves of Quercus liaotungensis Koidz. in Lingkong Mountain. Shanxi Province J Beijing for Univery 27:10–16. https://doi.org/10.3321/j.issn:1000-1522.2005.05.002. (in Chinese)
Li XP, Yu CY, Wu YY, Hong ZY, Sun J, Chen YP, Miao LX (2010) The biological reason for endangerment of Carpinus putoensis and measures for gene conservation. Scientia Silvae Sinicae 46:69–76. https://doi.org/10.11707/j.1001-7488.20100711. (in Chinese)
Li DS, Shi ZM, Feng QH, Liu F (2013) Response of leaf morphometric traits of Quercus species to climate in the temperate zone of the North-South Transect of Eastern China. Chinese J Plant Ecol 37:793–802. https://doi.org/10.3724/SP.J.1258.2013.00083. (in Chinese)
Li YG, Liu XH, Ma JW, Zhang XM, Xu LA (2018a) Phenotypic variation in Phoebe bournei populations preserved in the primary distribution area. J Forestry Res 29:35–44. https://doi.org/10.1007/s11676-017-0409-4
Li YH, Zhen Li, Xin ZM, Liu MH, Li YL, Hao YG (2018b) Effects of leaf shape plasticity on leaf surface temperature. Chinese J Plant Ecol 42:202–208. https://doi.org/10.17521/cjpe.2017.0127. (in Chinese)
Liao GL, Xu XB, Huang CH, Zhong M, Jia DF (2021) Resource evaluation and novel germplasm mining of Actinidia eriantha. Sci Hortic 282:110037. https://doi.org/10.1016/j.scienta.2021.110037
Lin Y, Liu S, Luo J, Liu X, Lu W, Wang C, Arnold RJ (2017) Landrace origins and phenotypic diversity through seedling morphology in Corymbia citriodora subsp. citriodora. Aust for 80:43–56. https://doi.org/10.1080/00049158.2016.1275494
Lister C, Dean C (1993) Recombinant inbred lines for mapping RFLP and phenotypic markers in Arabidopsis thaliana. Plant J 4:745–750. https://doi.org/10.1046/j.1365-313X.1993.04040745.x
Liu YJ, Zhang LR, Xu XL, Niu HS (2016) Understanding the wide geographic range of a clonal perennial grass: plasticity versus local adaptation. AoB Plants 8:141. https://doi.org/10.1093/aobpla/plv141
Lu ZQ (2017) Species delimitation in the subfamily Coryloideae of Betulaceae in China. PhD thesis, Lanzhou University, Lanzhou, China. https://doi.org/10.7666/d.D01297590 (in Chinese)
Maximowicz CJ (1881) Diagnoses plantarum novarum asiaticarum. IV Bulletin de l'Académie impériale des sciences de St.-Pétersbourg 27:425–560
McDonald JE (1962) Collection and washout of airborne pollens and spores by raindrops. Science 135:435–437. https://doi.org/10.1126/science.135.3502.435
Meng H, Wei X, Franklin SB, Wu H, Jiang M (2017) Geographical variation and the role of climate in leaf traits of a relict tree species across its distribution in China. Plant Biol 19:552–561. https://doi.org/10.1111/plb.12564
Milligan BG, Leebens-Mack J, Strand AE (2008) Conservation genetics: beyond the maintenance of marker diversity. Mol Ecol 3:423–435. https://doi.org/10.1111/j.1365-294X.1994.tb00082.x
Niinemets Ü, Al Afas N, Cescatti A, Pellis A, Ceulemans R (2004) Petiole length and biomass investment in support modify light interception efficiency in dense poplar plantations. Tree Physiol 24:141–154. https://doi.org/10.1093/treephys/24.2.141
Niklas KJ (1985) The aerodynamics of wind pollination. Bot Rev 51:328–386. https://doi.org/10.1007/BF02861079
Nobel PS (2005) Physicochemical and Environmental Plant Physiology. Elsevier, Amsterdam. https://doi.org/10.1016/B978-0-12-374143-1.X0001-4
Parkhurst DF, Loucks OL (1972) Optimal leaf size in relation to environment. J Ecol 60:505–537. https://doi.org/10.2307/2258359
Pigliucci M, Murren CJ, Schlichting CD (2006) Phenotypic plasticity and evolution by genetic assimilation. J Exp Biol 209:2362–2367. https://doi.org/10.1242/jeb.02070
Ritchie ME, Olff H (1999) Spatial scaling laws yield a synthetic theory of biodiversity. Nature 400:557–560. https://doi.org/10.1038/23010
Scarano D, Rubio F, Ruiz JJ, Rao R, Corrado G (2014) Morphological and genetic diversity among and within common bean (Phaseolus vulgaris L.) landraces from the Campania region (Southern Italy). Sci Hortic 180:72–78. https://doi.org/10.1016/j.scienta.2014.10.013
Smith WK, Geller GN (1980) Leaf and environmental parameters influencing transpiration: Theory and field measurements. Oecologia 46:308–313. https://doi.org/10.1007/BF00346257
Takenaka A (1994) Effects of leaf blade narrowness and petiole length on the light capture efficiency of a shoot. Ecol Res 9:109–114. https://doi.org/10.1007/BF02347485
Traiser C, Klotz S, Uhl D, Mosbrugger V (2005) Environmental signals from leaves - a physiognomic analysis of European vegetation. New Phytol 166:465–484. https://doi.org/10.1111/j.1469-8137.2005.01316.x
Vendramini F, Díaz S, Gurvich DE, Wilson PJ, Thompson K, Hodgson JG (2002) Leaf traits as indicators of resource-use strategy in floras with succulent species. New Phytol 154:147–157. https://doi.org/10.1046/j.1469-8137.2002.00357.x
Wang XR, Xie CP, Yi XG, Xiang QB (2007) Study on the morphological variations of Cerasus subhirtella var. ascendens in different populations. Bull Bot Res 27:746–752. https://doi.org/10.3969/j.issn.1673-5102.2007.06.018(inChinese)
Weijschedé J, Martínková J, de Kroon H, Huber H (2006) Shade avoidance in Trifolium repens: costs and benefits of plasticity in petiole length and leaf size. New Phytol 172:655–666. https://doi.org/10.1111/j.1469-8137.2006.01885.x
Westoby M, Falster DS, Moles AT, Vesk PA, Wright IJ (2002) Plant ecological strategies: Some leading dimensions of variation between species. Annu Rev Ecol Evol Syst 33:125–159. https://doi.org/10.1146/annurev.ecolsys.33.010802.150452
Wise RR, Olson AJ, Schrader SM, Sharkey TD (2004) Electron transport is the functional limitation of photosynthesis in field-grown Pima cotton plants at high temperature. Plant Cell Environ 27:717–724. https://doi.org/10.1111/j.1365-3040.2004.01171.x
Yang XY, Wang ZF, Luo WC, Guo XY, Zhang CH, Liu JQ, Ren GP (2019) Plastomes of Betulaceae and phylogenetic implications. J Syst Evol 57:508–518. https://doi.org/10.1111/jse.12479
Yang XY, Wang ZF, Zhang L, Hao GQ, Liu JQ, Yang YZ (2020) A chromosome-level reference genome of the hornbeam Carpinus Fangiana. Sci Data 7:24. https://doi.org/10.1038/s41597-020-0370-5
Yin J, Ahn HS, Ha SY, Hwang IH, Yoon KD, Chin YW, Lee MW (2018) Anti-skin ageing effects of phenolic compounds from Carpinus tschonoskii. Nat Prod Res 33:3317–3320. https://doi.org/10.1080/14786419.2018.1497026
Zhang ZZ, Ji MC, Fan YR, Zheng G, Liu ZG (2016) Flowering Properties and Pollen Viability of Carpinus tientaiensis. J Zhejiang for Sci Tech 36:10–13. https://doi.org/10.3969/j.issn.1001-3776.2016.01.003. (In Chinese)
Zhang ZY, Jin GZ, Liu ZL (2021) Effects of needle age on leaf traits and their correlations of Pinus koraiensis across different regions. Chin J Plant Ecol 45:253–264. https://doi.org/10.17521/cjpe.2020.0352. (in Chinese)
Zhu YR, Gong YB (2017) Methods of wind pollination. Biodiversity Sci 25:864–873. https://doi.org/10.17520/biods.2017069. (in Chinese)
Zhu JY, Zhang LF, Shen P, Ren BQ, Liang Y, Chen ZD (2014) Wind pollination characteristics of styles in Betulaceae. Chin Bull Bot 49:524–538. https://doi.org/10.3724/SP.J.1259.2014.00524. (in Chinese)
Zhu H, Zhu SX, Li YF, Yi XG, Duan YF, Wang XR (2018) Leaf phenotypic variation in natural populations of Cerasus dielsiana. Chin J Plant Ecol 42:1168–1178. https://doi.org/10.17521/cjpe.2018.0196. (in Chinese)
Zingaretti LM, Monfort A, Pérez-Enciso M (2021) Automatic fruit morphology phenome and genetic analysis: An application in the octoploid strawberry. Plant Phenomics. 2021:9812910. https://doi.org/10.34133/2021/9812910
Zwieniecki MA, Boyce CK, Holbrook NM (2004) Hydraulic limitations imposed by crown placement determine final size and shape of Quercus rubra L. leaves. Plant Cell Environ 27:357–365. https://doi.org/10.1111/j.1365-3040.2003.01153.x
Author information
Authors and Affiliations
Contributions
RZ designed the experiments and wrote the paper; RZ, XC and WL measured experimental data; RZ and QH collected experimental materials; ZZ guided research methods and ideas.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This research was supported by the Forestry and Grassland Science and Technology Achievements National Promotion Project of the National Forestry and Grassland Administration (2019133119), the Postgraduate Research and Practice Innovation Program of Jiangsu Province (KYCX22_1104), and the National Natural Science Foundation of China (31770752).
The online version is available at https://link.springer.com.
Corresponding editor: Yanbo Hu.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, R., Chu, X., He, Q. et al. Leaf phenotypic variation in natural populations of Carpinus tschonoskii in China. J. For. Res. 34, 1591–1602 (2023). https://doi.org/10.1007/s11676-022-01584-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-022-01584-0