Abstract
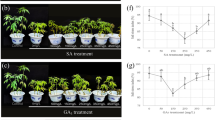
The changes in lipid peroxidation and the involvement of the antioxidant system in relation to salt stress tolerance were investigated in the callus of Acanthophyllum glandulosum and Acanthophyllum sordidum. The callus was subjected to NaCl stress (50–200 mM) for 40 d. The callus of A. glandulosum was less sensitive to NaCl stress than that of A. sordidum. Increasing concentrations of NaCl from 50 to 200 mM correlated to increased proline content in A. glandulosum. Total protein content was higher in extracts of A. glandulosum than in extracts of A. sordidum under both control and salinity treatments. Compared with A. sordidum, lipid peroxidation and H2O2 content were lower and the activities of superoxide dismutase (SOD), catalase (CAT), guaiacol peroxidase (GPX), ascorbate peroxidase, and glutathione reductase were higher in A. glandulosum under salt stress. Activity staining of antioxidant enzymes separated by native polyacrylamide gel electrophoresis (PAGE) revealed that callus of A. sordidum had five Fe-SOD isoforms and one Mn-SOD isoform, all of which were reduced by salinity. In A. glandulosum, two Mn-SOD, three Fe-SOD, and one Cu/Zn-SOD isoforms were detected. Out of these six SOD isoforms, expression of the Mn-SOD and Fe-SOD isoforms was enhanced at 100 mM and higher NaCl concentrations. Two and six GPX isoforms were detected in A. sordidum and A. glandulosum, respectively. Expression of the single CAT isoform in A. sordidum was preferentially reduced by salinity. In A. glandulosum, the two CAT isoforms showed differential down regulation under NaCl stress, with the CAT2 isoform detected only under control condition. These results suggest that A. glandulosum callus is better protected against salinity-induced oxidative damage by maintaining higher activities of antioxidant enzymes than the callus of A. sordidum.









Similar content being viewed by others
References
Aebi H. Catalases. In: Bergmeyer H. U. (ed) Methods of enzymatic analysis, vol. 2. Academic Press, New York, pp 673–684; 1974.
Abed Alrahman N. M.; Shibli R. A.; Ereifej K. I.; Hindiyeh M. Y. Influence of salinity on growth and physiology of in vitro grown cucumber (Cucumis sativus L.). Jordan J Agric Sci 1: 93–106; 2005.
Aghaleh M.; Niknam V.; Ebrahimzadeh H.; Razavi K. Salt stress effects on growth, pigments, proteins and lipid peroxidation in Salicornia persica and S. europaea. Biol Plant 53: 243–248; 2009.
Amor N. B.; Hamed K. B.; Debez A.; Grignon C.; Abdelly C. Physiological and antioxidant responses of the perennial halophyte Crithmum maritimum to salinity. Plant Sci 168: 889–899; 2005.
Ashraf M.; Harris P. J. C. Potential biochemical indicators of salinity tolerance in plants. Plant Sci 166: 3–16; 2004.
Azevedo Neto A. D.; Prisco J. T.; Eneas-Filho J.; de Abreu C. E. B.; Gomes-Filho E. Effect of salt stress on antioxidative enzymes and lipid peroxidation in leaves and roots of salt-tolerant and salt-sensitive maize genotypes. Environ Exp Bot 5: 87–94; 2006.
Azooz M. M.; Shaddad M. A.; Abdel-Latef A. A. The accumulation and compartmentation of proline in relation to salt tolerance of three sorghum cultivars. Indian J Plant Physiol 9: 1–8; 2004.
Bates L. S.; Waldern R. P.; Teare I. D. Rapid determination of free proline for water stress studies. Plant Soil 39: 205–207; 1973.
Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254; 1976.
Beauchamp C.; Fridovich I. Superoxide dismutase improved assays and an assay applicable to acrylamide gels. Anal Biochem 44: 276–287; 1971.
Bor M.; Őzdemir F.; Türkan I. The effect of salt stress on lipid peroxidation and antioxidants in leaves of sugar beet Beta vulgaris L. and wild beet Beta maritima L. Plant Sci 164: 77–84; 2003.
Carretero C. L.; Cantos M.; García J. L.; Troncoso A. In vitro–ex vitro salt (NaCl) tolerance of cassava (Manihot esculenta Crantz) plants. In Vitro Cell Dev Biol-Plant 43: 364–369; 2007.
Charbaji T.; Ayyoubi Z. Differntial growth of some grapevine varieties in Syria in response to salt in vitro. In Vitro Cell Dev Biol-Plant 40: 221–224; 2004.
Demiral T.; Türkan I. Comparative lipid peroxidation, antioxidant defense systems and proline content in roots of two rice cultivars differing in salt tolerance. Environ Exp Bot 53: 247–257; 2005.
Dewir Y. H.; Chakrabarty D.; Ali M. B.; Hahn E. J.; Paek K. Y. Lipid peroxidation and antioxidant enzyme activities of Euphorbia millii hyperhydric shoots. Environ Exp Bot 58: 93–99; 2006.
Ehrendorfer F. Polyploidy and distribution. In: Lews W. H. (ed) Polyploidy: biologia relevance. Plenum, New York, pp 45–60; 1980.
Fielding J. L.; Hall J. L. A biochemical and cytological study of peroxidase activity in roots of Pisum sativum. J Exp Bot 29: 969–981; 1978.
Gaidi G.; Miyamoto T.; Ramezani M.; Lacaille-Dubois M. A.; Glandulosides A.-D. Triterpene saponins from Acanthophyllum glandulosum. J Nat Prod 67: 1114–1118; 2004.
Ghaffari S. M. Cytotaxonomy of some species of Acanthophyllum (Caryophyllaceae) from Iran. Biol Bratislava 59: 53–60; 2004.
Heath R. L.; Packer L. Photoperoxidarion in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125: 189–198; 1968.
Al-Khayri J. M. Growth, proline accumulation and ion content in sodium chloride-stressed callus of date palm. In Vitro Cell Dev Biol-Plant 38: 79–82; 2002.
Jebara S.; Jebara M.; Limam F.; Elarbi Aouani M. Changes in ascorbate peroxidase, catalase, guaiacol peroxidase and superoxide dismutase activities in common bean (Phaseolus vulgaris) nodules under salt stress. J Plant Physiol 162: 929–936; 2005.
Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685; 1970.
Lin C. C.; Hsu Y. T.; Kao C. H. The effect of NaCl on proline accumulation in rice leaves. Plant Growth Regul 36: 275–285; 2002.
Lin C. C.; Kao C. H. NaCl induced changes in ionically bounds peroxidase activity in roots of rice seedlings. Plant Soil 216: 147–53; 1999.
Masood A.; Ahmad S. N.; Zeeshan M.; Abraham G. Differential response of antioxidant enzymes to salinity stress in two varieties of Azolla (Azolla pinnata and Azolla filiculoides). Environ Exp Bot 58: 216–222; 2006.
Meratan A. A.; Ghaffari S. M.; Niknam V. Effects of salinity on growth, proteins and antioxidant enzymes in three Acanthophyllum species of different ploidy levels. JSUT 33: 1–8; 2008.
Meratan A. A.; Ghaffari S. M.; Niknam V. In vitro organogenesis and antioxidant enzymes activity in Acanthophyllum sordidum. Biol Plant 53: 5–10; 2009.
McCoy T. J. Tissue culture evaluation of NaCl tolerance in Medicago sativa species: cellular versus whole plant response. Plant Cell Rep 6: 31–34; 1987.
Meloni D. A.; Oliva M. A.; Martinez C. A.; Cambraia J. Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ Exp Bot 49: 69–76; 2003.
Munns R.; Rebetzke G. J.; Husain S.; James R. A.; Hare R. A. Genetic control of sodium extrusion in durum wheat. Austra J Agr Res 54: 627–635; 2003.
Murashige T.; Skoog F. A revised medium for rapid growth and bioassay with tobacco tissue culture. Physiol Plant 15: 473–497; 1962.
Niknam V.; Bagherzadeh M.; Ebrahimzadeh H.; Sokhansanj A. Effect of NaCl on biomass and content of sugars, proline and proteins in seedlings and leaf explants of Nicotiana tabacum grown in vitro. Biol Plant 48: 613–615; 2004.
Niknam V.; Razavi N.; Ebrahimzadeh H.; Sharifizadeh B. Effect of NaCl on biomass, protein and proline contents, and antioxidant enzymes in seedlings and calli of two Trigonella species. Biol Plant 50: 591–596; 2006.
Noctor G.; Foyer C. H. Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49: 249–279; 1998.
Piqueras A.; Hernández J. A.; Olmos E.; Hellín E.; Sevilla F. Changes in antioxidant (CT) enzymes and organic solutes associated with adaptation of citrus cells to salt stress. Plant Cell Tiss Org Cul 45: 53–60; 1996.
Rao M. V.; Paliyath G.; Ormrod D. P. Ultraviolet-B- and ozone-induced biochemical changes in antioxidant enzymes of Arabidopsis thaliana. Plant Physiol 110: 125–136; 1996.
Sairam R. K.; Srivastava G. C. Changes in antioxidant activity in subcellular fractions of tolerant and susceptible wheat genotypes in response to long term salt stress. Plant Sci 162: 897–904; 2002.
Singh M.; Jaiswal U.; Jaiswal V. S. In vitro selection of NaCl-tolerant callus line and regeneration of plantlets in a Bamboo (Dendrocalamus strictus Needs.). In Vitro Cell Dev Biol-Plant 39: 229–233; 2003.
Soltis D. E.; Soltis P. S.; Tate J. A. Advances in the study of polyploidy since plant speciation. New Phytologist 161: 173–191; 2003.
Sudhakar C.; Lakshmi A.; Giridarakumar S. Changes in the antioxidant enzyme efficacy in two high yielding genotypes of mulberry (Morus alba L.) under NaCl salinity. Plant Sci 161: 613–619; 2001.
Vaidyanathan H.; Sivakumar P.; Chakrabarty R.; Thomas G. Scavenging of reactive oxygen species in NaCl-stressed rice (Oryza sativa L.)—differential response in salt-tolerant and sensitive varieties. Plant Sci 165: 1411–1418; 2003.
Velikova V.; Yordanov I.; Edreva A. Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci 151: 59–66; 2000.
Wahid A.; Masood I.; Javed I. U. H. Phenotypic flexibility as marker of sodium chloride tolerance in sunflower genotypes. Environ Exp Bot 42: 85–94; 1999.
Woodbury W.; Spencer A. K.; Stahmann M. A. An improved procedure using ferricyanide for detecting catalase isozymes. Anal Biochem 44: 301–305; 1971.
Zhang F.; Yang Y. L.; He W. L.; Zhao X.; Zhang X. Effects of salinity on growth and compatible solutes of callus induced from Populus euphratica. In Vitro Cell Dev Biol-Plant 40: 491–494; 2004.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: N.J. Taylor
Rights and permissions
About this article
Cite this article
Niknam, V., Meratan, A.A. & Ghaffari, S.M. The effect of salt stress on lipid peroxidation and antioxidative enzymes in callus of two Acanthophyllum species. In Vitro Cell.Dev.Biol.-Plant 47, 297–308 (2011). https://doi.org/10.1007/s11627-011-9345-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-011-9345-8