Abstract
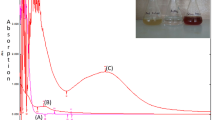
Compared with cultivated plants, wild weeds are cheaper and wider available for phytogenetic synthesis of nanoparticles. It is very valuable to distinguish the weed species from the local dominant wild plants for both efficient synthesis of nanoparticles and real industrial applications. In this study, a cheap, simple and novel biosynthesis method for silver nanoparticles (AgNPs) using Chenopodium aristatum. L stem extract as reducing and capping agents was developed. The real-time formation of AgNPs was monitored by UV–vis spectroscopy, and the SPR band was located at ~440 nm. The reaction factors including incubation time, AgNO3 concentration, reaction temperature and pH were discussed and optimized. The synthesized AgNPs were quasi-spherical with the sizes from 3 to 36 nm. HRTEM images and XRD patterns showed that the AgNPs were with the face-centered cubic (fcc) structure. FTIR spectra indicated that some functional groups such like O–H, N–H, C–H, C=O were possibly responsible for reducing silver ions into nanoparticles and acted as capping agents to avoid agglomeration. The synthesized AgNPs exhibited strong catalytic activity in degradation of 4-nitrophenol and good antibacterial activity against E. coli and S. aureus. The proposed phytogenetic synthesis method is economical, simple and feasible.










Similar content being viewed by others
References
Q. Chaudhry and L. Castle (2011). Trends. Food. Sci. Tech. 22, 595–603.
S. Ahmed, M. Ahmad, B. L. Swami, and S. Ikram (2016). J. Adv. Res. 7, 17–28.
G. Martínez-Castañón, F. Martínez-Gutierrez, J. Martínez-Mendoza, and F. Ruiz (2008). J. Nanopart. Res. 10, 1343–1348.
C. Larue, H. Castillo-Michel, S. Sobanska, L. Cécillon, S. Bureau, V. Barthès, L. Ouerdane, M. Carrière, and G. Sarret (2014). J. Hazard. Mater. 264C, 98–106.
P. Singh, Y. J. Kim, D. Zhang, and D. C. Yang (2016). Trends. Biotechnol. 34, 588–599.
R. Rajan, K. Chandran, S. L. Harper, S. I. Yun, and P. T. Kalaichelvan (2015). Ind. Crop. Prod. 70, 356–373.
M. Apte, P. Chaudhari, A. Vaidya, A. R. Kumar, and S. Zinjarde (2016). Colloid Surf. A501, 1–8.
S. A. Annu, S. Ikram, and Y. S. Salprima (2016). J. Phototoch. Photobio. B 161, 141–153.
P. Dauthal and M. Mukhopadhyay (2016). Ind. Eng. Chem. Res. 55, 9557–9577.
I. M. Chung, I. Park, K. Seunghyun, M. Thiruvengadam, and G. Rajakumar (2016). Nanoscale. Res. Lett. 11, 1–14.
K. Kalishwaralal, V. Deepak, S. B. R. K. Pandian, M. Kottaisamy, S. Barathmanikanth, B. Kartikeyan, and S. Gurunathan (2010). Colloid Surf. B 77, 257–262.
P. Nalawade, P. Mukherjee, and S. Kapoor (2014). Spectrochim. Acta. A 129C, 121–124.
U. B. Jagtap and V. A. Bapat (2013). Ind. Crop. Prod. 46, 132–137.
S. Kaviya, J. Santhanalakshmi, B. Viswanathan, J. Muthumary, and K. Srinivasan (2011). Spectrochim. Acta. A 79, 594–598.
M. Ghaffari-Moghaddam and R. Hadi-Dabanlou (2014). J. Ind. Eng. Chem. 20, 739–744.
P. P. N. V. Kumar, S. V. N. Pammi, P. Kollu, K. V. V. Satyanarayana, and U. Shameem (2014). Ind. Crop. Prod. 52, 562–566.
M. Sathishkumar, K. Sneha, I. S. Kwak, J. Mao, S. J. Tripathy, and Y. S. Yun (2009). J. Hazard. Mater. 171, 400–404.
S. Ghosh, S. Patil, M. Ahire, R. Kitture, A. Jabgunde, S. Kale, K. Pardesi, J. Bellare, D. D. Dhavale, and B. A. Chopade (2011). J. Nanomater. 2011, 18083–18088.
D. Philip and C. Unni (2011). Physica E 43, 1318–1322.
M. M. H. Khalil, E. H. Ismail, and F. El-Magdoub (2012). Arab. J. Chem. 5, 431–437.
D. Philip (2010). Physica E 42, 1417–1424.
A. Nabikhan, K. Kandasamy, A. Raj, and N. M. Alikunhi (2010). Colloid Surf. B79, 488–493.
C. S. Espenti, K. S. V. K. Rao, and K. M. Rao (2016). Mater. Lett. 174, 129–133.
J. L. López-Miranda, M. Vázquez, N. Fletes, R. Esparza, and G. Rosas (2016). Mater. Lett. 176, 285–289.
A. Verma and M. S. Mehata (2015). J. Radiat. Res. Appl. Sci. 9, 109–115.
V. Ravichandran, S. Vasanthi, S. Shalini, S. A. A. Shah, and R. Harish (2016). Mater. Lett. 180, 264–267.
M. J. Ahmed, G. Murtaza, A. Mehmood, and T. M. Bhatti (2015). Mater. Lett. 153, 10–13.
S. Ahmed, S. Ullah, M. Ahmad, B. L. Swami, and S. Ikram (2016). J. Radiat. Res. Appl. Sci. 9, 1–7.
B. Paul, B. Bhuyan, D. D. Purkayastha, and S. S. Dhar (2016). J. Phototoch Photobio. B 154, 1–7.
N. Khatoon, R. Ahmad, and M. Sardar (2015). Biochem. Eng. J. 102, 91–97.
M. A. Farah, M. A. Ali, S. M. Chen, Y. Li, F. M. Al-Hemaid, F. M. Abou-Tarboush, K. M. Al-Anazi, and J. Lee (2016). Colloid Surf. B 141, 158–169.
R. A. Morales-Luckie, A. A. Lopezfuentes-Ruiz, O. F. Olea-Mejía, A. F. Liliana, V. Sanchez-Mendieta, W. Brostow, and J. P. Hinestroza (2016). Mater. Sci. Eng. C 69, 429–436.
V. Dhand, L. Soumya, S. Bharadwaj, S. Chakra, D. Bhatt, and B. Sreedhar (2016). Mater. Sci. Eng. C 58, 36–43.
S. Mohammadi and S. Pourseyedi (2016). J. Environ. Chem. Eng. 4, 2023–2032.
J. K. Patra, G. Das, and K. H. Baek (2016). J. Phototoch. Photobio. B 161, 200–210.
B. Kumar, K. Smita, L. Cumbal, J. Camacho, E. Hernández-Gallegos, M. Grijalva, and K. Andrade (2016). Mater. Sci. Eng. C 62, 725–731.
N. H. Rao, N. Lakshmidevi, S. V. N. Pammi, P. Kollu, S. Ganapaty, and P. Lakshmi (2016). Mater. Sci. Eng. C 62, 553–557.
D. Nayak, S. Ashe, P. R. Rauta, M. Kumari, and B. Nayak (2016). Mater. Sci. Eng. C 58, 44–52.
T. N. Edison, Y. R. Lee, and M. G. Sethuraman (2016). Spectrochim. Acta. A 161, 122–129.
C. Krishnaraj, E. G. Jagan, S. Rajasekar, P. Selvakumar, P. T. Kalaichelvan, and N. Mohan (2010). Colloid Surf. B 76, 50–56.
H. Alishah, S. P. Seyedi, S. Y. Ebrahimipour, and S. Esmaeili-Mahani (2016). J. Clust. Sci. 27, 421–429.
T. N. J. I. Edison, E. R. Baral, Y. R. Lee, and S. H. Kim (2016). J. Clust. Sci. 27, 285–298.
S. C. Bankalgi, R. L. Londonkar, U. Madire, and N. K. A. Tukappa (2016). J. Clust. Sci. 27, 1485–1497.
M. Khatami, R. Mehnipor, M. H. S. Poor, and G. S. Jouzani (2016). J. Clust. Sci. 27, 1601–1612.
J. Venugobal and K. Anandalakshmi (2016). J. Clust. Sci. 27, 1683–1699.
Y. G. Yin, X. Y. Yang, L. G. Hu, Z. Q. Tan, L. X. Zhao, Z. Zhang, J. F. Liu, and G. B. Jiang (2016). Environ. Sci. Technol. Lett. 3, (4), 160–165.
F. Qazi, Z. Hussain, and M. N. Tahir (2016). RSC Adv. 6, 60277–60286.
S. S. Shankar, A. Ahmad, R. Pasricha, M. I. Khan, R. Kumar, and M. Sastry (2004). J. Colloid. Inter. Sci. 274, 69–75.
S. J. Yu, Y. G. Yin, X. X. Zhou, L. J. Dong, and J. F. Liu (2016). Environ. Sci Nano. 3, 883–893.
A. Tripathy, A. M. Raichur, N. Chandrasekaran, and T. C. Prathna (2010). J. Nanopart. Res. 12, 237–246.
M. Ahamed, M. A. M. Khan, M. K. J. Siddiqui, M. S. Alsalhi, and S. A. Alrokayan (2011). Physica E 43, 1266–1271.
S. Link and M. A. El-Sayed (1999). J. Phys. Chem. B 103, 4212–4217.
N. Muniyappan and N. S. Nagarajan (2014). Process Biochem 49, 1054–1061.
C. Tamuly, M. Hazarika, M. Bordoloi, P. K. Bhattacharyya, and R. Kar (2014). Spectrochim. Acta A 132, 687–691.
Acknowledgements
This work was kindly co-funded by the National Natural Science Foundation of China (21277043, 21620102008), the Beijing Natural Science Foundation (8132038), the Foundation of State Key Laboratory of Coal Combustion (FSKLCC1414) and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yuan, CG., Huo, C., Gui, B. et al. Green Synthesis of Silver Nanoparticles Using Chenopodium aristatum L. Stem Extract and Their Catalytic/Antibacterial Activities. J Clust Sci 28, 1319–1333 (2017). https://doi.org/10.1007/s10876-016-1147-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-016-1147-z