Abstract
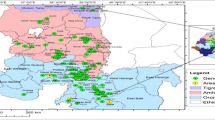
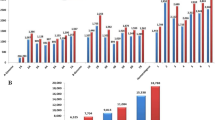
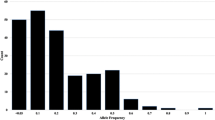
Genetic diversity is an important resource to improve new wheat cultivars in the breeding program. In this study, fluorescence in situ hybridization (FISH) and genotyping by sequencing (GBS) were used to evaluate the genetic diversity and population structure of the Timopheevii group (AtAtGG, 2n = 4x = 28) including 15 Triticum timopheevii Zhuk. accessions and 35 Triticum araraticum Jakubz. accessions. FISH analysis showed that there were 14 and 19 FISH signal variations in the At and G genome of T. araraticum, respectively. But there were only two signal variations in the G genome of T. timopheevii. In this study, 190,402 SNP markers were obtained from GBS, in which the lowest and highest frequencies of SNPs were found in the G and At genomes, respectively. Genetic diversity analysis of the 50 accessions indicated that the mean GD and PIC were 0.30 and 0.26, with the ranges of 0.1–0.5 and 0.1–0.4, respectively. The lowest and highest numbers of SNPs identified on the chromosomes 4G and 2At were 7748 and 17,527, respectively. Structure and cluster analyses divided 50 accessions into two subpopulations (POP1 and POP2). POP1 consisted of most T. araraticum accessions, and POP2 comprised all accessions of T. timopheevii and one T. araraticum accession AS273. In addition, the genetic variance showed that genetic variation was greater within populations (97%) than between populations (3%). The present results provided important information for the improvement of cultivated durum and hexaploid wheat production in future breeding programs of China and other countries.










Similar content being viewed by others
Availability of data and materials
The data that support the findings of this study are available in ‘figshare’ with the identifier data DOIs, including two datasets (Tetraploid wheat-GBS.vcf, https://doi.org/10.6084/m9.figshare.16692490.v1 and https://doi.org/10.6084/m9.figshare.17032322.v1).
References
Alexander DH, Novembre J, Lange K (2009) Fast model-based estimation of ancestry in unrelated individuals. Genome Res 19(9):1655–1664. https://doi.org/10.1101/gr.094052.109
Alipour H, Bihamta MR, Mohammadi V, Peyghambari SA, Bai G, Zhang G (2017) Genotyping-by-sequencing (GBS) revealed molecular genetic diversity of Iranian wheat landraces and cultivars. Front Plant Sci 8:1293. https://doi.org/10.3389/fpls.2017.01293
Arora A, Kundu S, Dilbaghi N, Sharma I, Tiwari R (2014) Population structure and genetic diversity among Indian wheat varieties using microsatellite (SSR) markers. Aust J Crop Sci 8:1281–1289. https://doi.org/10.3316/informit.675809051169521
Badaeva ED, Boguslavsky RL, Badaev NS, Zelenin AV (1990) Intraspecific chromosomal polymorphism of Triticum araraticum (Poaceae) detected by C-banding technique. Pl Syst Evol 169:13–24. https://doi.org/10.1007/BF00935980
Badaeva ED, Budashkina EB, Badaev NS, Kalinina NP, Shkutina FM (1991) General features of chromosome substitutions in Triticum aestivum × T. timopheevi hybrids. Theor Appl Genet 82:227–232. https://doi.org/10.1007/BF00226218
Badaeva ED, Filatenko AA, Badaev NS (1994a) Cytogenetic investigation of Triticum timopheevi (Zhuk.) Zhuk. and related species using C-banding. Theor Appl Genet 89:622–628. https://doi.org/10.1007/BF00222457
Badaeva ED, Badaev NS, Gill BS, Filatenko AA (1994b) Intraspecific karyotype divergence in Triticum araraticum (Poaceae). Pl Syst Evol 192:117–145. https://doi.org/10.1007/BF00985912
Badaeva ED, Ruban AS, Zoshchuk SA, Surzhikov SA, Knu¨pffer H, Kilian B, (2016) Molecular cytogenetic characterization of Triticum timopheevi chromosomes provides new insight on genome evolution of T. zhukovskyi. Plant Syst Evol 302:943–956. https://doi.org/10.1007/s00606-016-1309-3
Badaeva ED, Konovalov FA, Knüpffer H, Fricano A, Ruban AS, KeheZ ZSA, Surzhikov SA, Neumann K, Graner A, Hammer K, Filatenko A, Bogaard A, Jones G, Özkan H, Kilian B (2022) Genetic diversity, distribution and domestication history of the neglected GGAtAt genepool of wheat. Theor Appl Genet 135:755–776. https://doi.org/10.1007/s00122-021-03912-0
Barradas MC, Romano MCS (1979–1980) Hybrid wheat: I. Investigation of the capacity for outcrossing in Triticum aestivum subsp. vulgare. Melhoramento 28:117–126
Batley J, Edwards D (2007) SNP applications in plants. In: Oraguzie NC, Rikkerink EHA, Gardiner SE, De Silva HN (eds) Association Mapping in Plants. Springer, New York, pp 95–102. https://doi.org/10.1007/978-0-387-36011-9-6
Belea A (1992) Interspecific and intergeneric crosses in cultivated plants. Akademiai Kiado, Budapest, pp 54–66
Botstein D, White RL, Skolnick M, Davis RW (1980) Construction of a genetic linkage map in man using restriction fragment length polymorphisms. Am J Hum Genet 32:314–331. https://doi.org/10.1016/0165-1161(81)90274-0
Brown-Guedira GL, Badaeva ED, Gill BS, Cox TS (1996) Chromosome substitutions of Triticum timopheevii in common wheat and some observations on the evolution of polyploid wheat species. Theor Appl Genet 93:1291–1298. https://doi.org/10.1007/BF00223462
Brown-Guedira GL, Singh S, Fritz AK (2003) Performance and mapping of leaf rust resistance transferred to wheat from Triticum timopheevii subsp. armeniacum. Phytopathology 93:784–789. https://doi.org/10.1094/PHYTO.2003.93.7.784
Czajkowska BI, Bogaard A, Charles M, Jones G, Brown TA (2020) Ancient DNA typing indicates that the “new” glume wheat of early Eurasian agriculture is a cultivated member of the Triticum timopheevi group. J Archaeol Sci 123:105258. https://doi.org/10.1016/j.jas.2020.105258
Danecek P, Auton A, Abecasis G, Albers CA, Banks E, DePristo MA, Handsaker RE, Lunter G, Marth GT, Sherry ST, McVean G, Durbin R (2011) The variant call format and VCFtools. Bioinformatics 27:2156–2158. https://doi.org/10.1093/bioinformatics/btr330
De Moulins D (1993) Les restes des plantes carbonisées du Cafer Höyük. Cahiers De L’euphrate 7:191–234
Dorofeev VF, Filatenko AA, Migushova EF, Udachin RA, Jakubziner MM (1979) Cultivated flora of the USSR. In: Brezhnev DD (ed) Wheat, vol 1. Kolos (Leningrad branch), Leningrad (in Russian)
Dvorak JDI, Terlizzi P, Zhang HB, Resta P (1993) The evolution of polyploid wheats: identification of the A genome donor species. Genome 36:21–31. https://doi.org/10.1139/g93-004
Edet OU, Gorafi YSA, Nasuda S, Tsujimoto H (2018) DArTseq-based analysis of genomic relationships among species of tribe Triticeae. Sci Rep 8:16397–16397. https://doi.org/10.1038/s41598-018-34811-y
Elshire RJ, Glaubitz JC, Sun Q, Poland JA, Kawamoto K, Buckler ES et al (2011) A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 6:e19379. https://doi.org/10.1371/journal.pone.0019379
Golovnina K, Gluskov S, Blinov A, Myorov V, Adkison L, Goncharov N (2007) Molecular phylogeny of the genus Triticum L. Plant Syst Evol 264:195–216. https://doi.org/10.1007/s00606-006-0478-x
Gornicki P, Zhu H, Wang J, Challa GS, Zhang Z, Gill BS et al (2014) The chloroplast view of the evolution of polyploid wheat. New Phytol 204:704–714. https://doi.org/10.1111/nph.12931
Govindaraj M, Vetriventhan M, Srinivasan M (2015) Importance of genetic diversity assessment in crop plants and its recent advances: an overview of its analytical perspectives. Genet Res Intl 431487:1–14. https://doi.org/10.1155/2015/431487
Griffin WB (1987) Outcrossing in New Zealand wheats measured by occurrence of purple grain. New Zeal J Agric Res 30:287–290. https://doi.org/10.1080/00288233.1987.10421885
Hao M, Luo JT, Zhang LQ, Yuan ZW, Yang YW, Wu M et al (2013) Production of hexaploid triticale by a synthetic hexaploid wheat-rye hybrid method. Euphytica 193:347–357. https://doi.org/10.1007/s10681-013-0930-2
He J, Zhao X, Laroche A, Lu ZX, Liu HK, Li Z (2014) Genotypingby-sequencing (GBS), an ultimate marker-assisted selection (MAS) tool to accelerate plant breeding. Front Plant Sci 5:484. https://doi.org/10.3389/fpls.2014.00484
Huang S, Sirikhachornkit A, Su X, Faris J, Gill B, Haselkorn R et al (2002) Genes encoding plastid acetyl-CoA carboxylase and 3-phosphoglycerate kinase of the Triticum/Aegilops complex and the evolutionary history of polyploid wheat. Proc Natl Acad Sci USA 99:8133–8138. https://doi.org/10.1073/pnas.072223799
Huang YF, Poland JA, Wight CP, Jackson EW, Tinker NA (2014) Using genotyping-by-sequencing (GBS) for genomic discovery in cultivated oat. PLoS ONE 9:e102448. https://doi.org/10.1371/journal.pone.0102448
Hucl P (1996) Out-crossing rates for 10 Canadian spring wheat cultivars. Can J Plant Sci 76:423–427. https://doi.org/10.4141/cjps96-075
Hyun DY, Sebastin R, Lee KJ, Lee GA, Shin MJ, Kim SH et al (2020) Genotyping-by-sequencing derived single nucleotide polymorphisms provide the first well-resolved phylogeny for the genus Triticum (Poaceae). Front Plant Sci 11:688. https://doi.org/10.3389/fpls.2020.00688
Jakobashvili ZA (1989) Determination of phylogenetic relationships between wheat species by the analysis of polymorphism and inheritance of storage proteins. Ph.d thesis, Institute of General Genetics, the USSR Academy of Sciences (in Russian)
Jakubziner MM (1933) Natural growing places of wild wheat in USSR. Priroda 8–9:98–102 (in Russian)
Jakubziner MM (1947) Forms and species of hardy wheats. Selektsiya i 548 Semenovodstvo (Plant Breeding and Seed Growing) 14(5):46 (in Russian)
Jakubziner MM (1950) New data on the wild wheat Triticum araraticum Jakubc. Bull Appl Bot Genet Pl-Breed 28:199–200 (in Russian)
Jӓrve K, Jakobson I, Enno T (2002) Tetraploid wheat species Triticum timopheevi and Triticum militinae in common wheat improvement. Acta Agron Hung 50:463–477. https://doi.org/10.1556/AAgr.50.2002.4.9
Keydel F (1978) Floral biology and its influence on varietal maintenance and multiplication in autogamous cereals. Saatgutwirtschaft 30:246–250
Kilian B, Özkan H, Deusch O, Effgen S, Brandolini A, Kohl J et al (2007) Independent wheat B and G genome origins in outcrossing Aegilops progenitor haplotypes. Mol Biol Evol 24:217–227. https://doi.org/10.1093/molbev/msl151
Kilian B, Özkan H, Pozzi C, Salamini F (2009) Domestication of the Triticeae in the Fertile Crescent. In: Muehlbauer GJ, Feuillet C (eds) Genetics and genomics of the Triticeae. Springer, New York, pp 81–119. https://doi.org/10.1007/978-0-387-77489-3_3
King J, Grewal S, Othmeni M, Coombes B, Yang C, Walter N, Ashling S, Scholefield D, Walker J, Hubbart-Edwards S, Hall A, King IP (2022) Introgression of the Triticum timopheevii genome into wheat detected by chromosome-specific kompetitive allele specific PCR markers. Front Plant Sci 13:919519. https://doi.org/10.3389/fpls.2022.919519
Kumar S, Banks TW, Cloutier S (2012) SNP discovery through next generation sequencing and its applications. Int J Plant Genomics 2012:831460. https://doi.org/10.1155/2012/831460
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25:1754–1760. https://doi.org/10.1093/bioinformatics/btp324
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N et al (2009) The sequence alignment/map format and SAM tools. Bioinformatics 25(16):2078–2079. https://doi.org/10.1093/bioinformatics/btp352
Li R, Li CQ, Chen HY, Liu XH, Xiao H, Chen SY (2019) Genomic diversity and admixture patterns among six Chinese indigenous cattle breeds in Yunnan. Asian Australas J Anim Sci 32(8):1069–1076. https://doi.org/10.5713/ajas.18.0605
Li LF, Zhang ZB, Wang ZH, Li N, Sha Y, Wang XF et al (2022) Genome sequences of five Sitopsis species of Aegilops and the origin of polyploid wheat B subgenome. Mol Plant 15:488–503. https://doi.org/10.1016/j.molp.2021.12.019
Lilienfeld F, Kihara H (1934) Genomanalyze bei Triticum und Aegilops. V. Triticum timopheevi Zhuk. cytologia (Tokyo) 6:87–122. https://doi.org/10.1508/cytologia.6.87
Lilienfeld FA, Kihara H (1951) Genome-analysis in Triticum and Aegilops. X. Concluding Review. Cytologia 16:101–123
Liu K, Muse SV (2005) PowerMarker: an integrated analysis environment for genetic marker analysis. Bioinformatics 21:2128–2129. https://doi.org/10.1093/bioinformatics/bti282
Liu XJ, Zhang MH, Liu X, Li HY, Hao M, Ning SZ et al (2018) Molecular cytogenetic identification of newly synthetic Triticum kiharae with high resistance to stripe rust. Genet Resour Crop Evol 65(6):1725–1732. https://doi.org/10.1007/s10722-018-0649-y
Luo Z, Brock J, Dyer JM, Kutchan T, Schachtman D, Augustin M, Ge Y, Fahlgren N et al (2019) Genetic diversity and population structure of a Camelina sativa spring panel. Front Plant Sci 10:184. https://doi.org/10.3389/fpls.2019.00184
Martin TJ (1990) Outcrossing in 12 hard red winter-wheat cultivars. Crop Sci 30:59–62. https://doi.org/10.2135/cropsci1990.0011183X003000010013x
McIntosh RA, Gyárfás J (1971) Triticum timopheevi as a source of resistance to wheat stem rust. Z Pflflanzenzüchtg 66:240–248
Menabde VL, Ericzjan AA (1960) To the investigation of Georgian wheat Zanduri. Proc Georg Acad Sci 25:731–732 (in Georgian)
Mikó P, Megyeri M, Farkas A, Molnár I, Molnár-Láng M (2015) Molecular cytogenetic identification and phenotypic description of a new synthetic amphiploid, Triticum timococcum (AtAtGGAmAm). Genet Resour Crop Evol 62:55–66. https://doi.org/10.1007/s10722-014-0135-0
Mori N, Miyashita NT, Terachi T, Nakamura C (1997) Variation in coxII intron in the wild ancestral species of wheat. Hereditas 126:281–288. https://doi.org/10.1111/j.1601-5223.1997.00281.x
Mori N, Kondo Y, Ishii T, Kawahara T, Valkoun J, Nakamura C (2009) Genetic diversity and origin of timopheevi wheat inferred by chloroplast DNA fingerprinting. Breed Sci 59:571–578. https://doi.org/10.1270/jsbbs.59.571
Nave M, Ta M, Raupp J, Tiwari VK, Ozkan H, Poland J, Hale I, Komatsuda T, Distelfeld A (2021) The independent domestication of Timopheev’s wheat: insights from haplotype analysis of the Brittle rachis 1 (BTR1-A) gene. Gene 12:338. https://doi.org/10.3390/genes12030338
Nesbitt M, Samuel D (1996) From staple crop to extinction? The archaeology and history of the hulled wheats. In: Padulosi S, Hammer K, Heller J (eds) Hulled wheats. Proceedings of the first international workshop on hulled wheats. Castelvecchio Pascoli, Tuscany, Italy, pp 41–100
Okada T, Jayasinghe J, Nansamba M, Baes M, Warner P, Kouidri, et al (2018) Unfertilized ovary pushes wheat flower open for cross-pollination. J Exp Bot 3:399–412. https://doi.org/10.1093/jxb/erx410
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in excel, Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Peterson GW, Dong Y, Horbach C, Fu YB (2014) Genotyping-by sequencing for plant genetic diversity analysis: a lab guide for SNP genotyping. Diversity 6:665–680. https://doi.org/10.3390/d6040665
Peusha HO, Enno TM, Priilinn O (1996) Genetic analysis of disease resistance in wheat hybrids, derivatives of Triticum timopheevi and Triticum militinae. Acta Agron Hung 44:237–244
Poland JA, Brown PJ, Sorrells ME, Jannink J (2012) Development of high-density genetic maps for barley and wheat using a novel two-enzyme genotyping-by-sequencing approach. PLoS ONE 7:e32253. https://doi.org/10.1371/journal.pone.0032253
Rodríguez S, Maestra M, Perera E, Díez M, Naranjo T (2000) Pairing affinities of the B- and G-genome chromosomes of polyploid wheats with those of Aegilops speltoides. Genome 43:814–819. https://doi.org/10.1139/g00-055
Salem KFM, Sallam A (2016) Analysis of population structure and genetic diversity of Egyptian and exotic rice (Oryza sativa L.) genotypes. C R Biol 339:1–9. https://doi.org/10.1016/j.crvi.2015.11.003
Salina EA, Leonova IN, Efremova TT, Röder MS (2006) Wheat genome structure: translocations during the course of polyploidization. Funct Integr Genomics 6:71–80. https://doi.org/10.1007/s10142-005-0001-4
Shcherban AB, Schichkina AA, Salina EA (2016) The occurrence of spring forms in tetraploid Timopheevi wheat is associated with variation in the first intron of the VRN-A1 gene. BMC Plant Biol 16:107–118. https://doi.org/10.1186/s12870-016-0925-y
Timonova EM, Leonova IN, Röder MS, Salina EA (2013) Marker-assisted development and characterization of a set of Triticum aestivum lines carrying different introgressions from the T. timopheevii genome. Mol Breed 31:123–136. https://doi.org/10.1007/s11032-012-9776-x
Ulaş B, Fiorentino G (2021) Recent attestations of “new” glume wheat in Turkey: a reassessment of its role in the reconstruction of Neolithic agriculture. Veg Hist and Archaeobot 30:685–701. https://doi.org/10.1007/s00334-020-00807-w
Wagenaar EB (1966) Studies on the genome constitution of Triticum timopheevi Zhuk., II. The timopheevi complex and its origin. Evolution 20:150–164. https://doi.org/10.1111/j.1558-5646.1966.tb03351.x
Wright S (1965) The interpretation of population structure by F-statistics with special regard to systems of mating. Evolution 19:395–420. https://doi.org/10.1111/j.1558-5646.1965.tb01731.x
Yang X, Tan B, Liu H, Zhu W, Xu L et al (2020) Genetic diversity and population structure of Asian and European common wheat accessions based on genotyping-by-sequencing. Front Genet 11:580782. https://doi.org/10.3389/fgene.2020.580782
Zhang LQ, Yen Y, Zheng YL, Liu DC (2007) Meiotic restriction in emmer wheat is controlled by one or more nuclear genes that continue to function in derived lines. Sex Plant Reprod 20:159–166. https://doi.org/10.1007/s00497-007-0052-x
Zhukovsky PM (1924) Wild emmer in Georgia-Triticum dicoccum Schrank dicoccoides Koern. Notes Appl Sci Dept Tiflflis Bot Garden 3:1–3 (in Russian)
Zohary D, Hopf M, Weiss E (2012) Domestication of plants in the Old World, 4th edn. Oxford University Press, Oxford
Funding
This work was supported by the Key Research and Development Program of Sichuan Province, China (2021YFYZ0002) and the National Natural Science Foundation of China (91935303 and 31671682).
Author information
Authors and Affiliations
Contributions
TP and LQZ designed the research and wrote the manuscript. TP, XMJ, DHW, MHZ and XL performed the experiments. MH and XC helped with data analysis, and WL, BJ, LH, SZN, ZWY, DCL, BHW, ZHY and XJC supervised the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Human and animals rights
None.
Informed consent
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peng, T., Jiang, X., Wang, D. et al. Population structure and genetic diversity of Triticum araraticum Jakubz. and Triticum timopheevii Zhuk.. Genet Resour Crop Evol 70, 1799–1815 (2023). https://doi.org/10.1007/s10722-023-01537-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-023-01537-4