Abstract
The freshwater snail Pseudoplotia scabra Müller (Thiaridae) was first spotted in Lake Kinneret, Israel, in the mid 2000s. In a series of field surveys we followed its spread, documenting how by the end of 2010 this invasive mollusc formed >95 % of the snails in Lake Kinneret, nearly eradicating four native species.
Similar content being viewed by others
Introduction
Four species of freshwater snails have been abundant in the Jordan Valley and the Lake Kinneret (Sea of Galilee) region of Israel for the past 2 million years: Melanopsis costata (Melanopsidae), Melanoides tuberculata (Thiaridae), Theodoxus jordani (Neritidae) and Bithynia phialensis (Bithyniidae; Fig. 1), (Heller 2010). Up to about 1990 the first three of these four species were so common in Lake Kinneret that they covered every rock and boulder and every soft substrate, from the waterline to a depth of 15 m, with densities reaching hundreds of individuals m−2 (Dagan 1972; Tchernov 1975, J. Heller pers. obs.). By 2004, however, only very few live snails were found during two surveys at nine sites around the lake (J. Heller). In a dramatic shift during the period 2007–2009 large quantities of empty Pseudoplotia scabra (Müller, 1774) (Thiaridae) (Fig. 1) shells were collected along the southern shores of the lake, including 6,400 empty shells in 1 m2 during one visit in 2008 (Mienis and Mienis 2008). The present note documents the large change that occurred to the snail population in Lake Kinneret with the first known major invasion of a snail in at least the past 5,000 years (based on abundant snail shells in archaeological excavations; T. Zohary unpublished data). Based on a series of surveys we document the rapid domination of the lake’s snail community by Pseudoplotia scabra population. This species is parthenogenetic (personal observations). Pseudoplotia scabra was formerly placed in the genus Thiara by various authors (see Glaubrecht et al. 2009) but in Pseudoplotia by Mienis (2012), followed here.
The invasive snail Pseudoplotia scabra (apertural and abapertural views) and four native snails once abundant in Lake Kinneret: Melanopsis costata, Melanoides tuberculata, Theodoxus jordani and Bithynia phialensis. Scale bars are 5 mm. (The drawing of P. scabra is original, others are from Heller (2010), Courtesy Pensoft)
Methods
While the focus of this note is a series of surveys conducted in Lake Kinneret over the course of 2010–2011 we include some results from an additional survey conducted in 2004 and observations made in 2009. The merging of the results provides a more complete picture of the P. scabra invasion. The surveys included sampling of shallow sites (0–2 m) located in the southern, western and northern parts of the lake in 2004. Nine surveys were conducted over the course of 2010–2011, the first was terrestrial (piles of shells distant from the waterline); the other eight were aquatic. Between 5 and 19 sites were sampled on each aquatic survey and 2–5 depths were sampled at each site. Due to the large differences in substrate at the various sampling sites a range of sampling techniques were used. As such, we limit our report mainly to the relative contribution of P. scabra at the different sites and quantitatively compare densities at different times only at two sites for which the sampling technique was consistent over time. Voucher specimens of P. scabra from Lake Kinneret are deposited in the Mollusc Collection of the Hebrew University, Jerusalem (HUJ 22344).
Results
November 2004: In the southern and western part of the lake no snails were found. Similarly, at most sites in the north-eastern part of the lake no snails were found, with just 11 live M. tuberculata found at one site and one live M. costata at another. P. scabra was not seen.
Summer 2009: Live snails and empty shells of P. scabra were observed along four shores in southern and south-western parts of the lake but not in two other sites, north of Tiberias. Observations were not quantified and the eastern shores were not examined.
January 2010 (terrestrial): In piles of empty shells distant from the waterline at the south-eastern part of the lake many P. scabra shells were found. No shells of this species were found in shell piles at nine other sites around the lake.
February 2010 (12 sites; 13 samples; 0.2 and 0.8 m depth): Live P. scabra were found only in two samples; no other live snails were found in those two samples. A few individuals of Melanopsis costata and Theodoxus jordani were found, in three of the samples. In eight samples no snails were found.
March 2010 (17 sites; 32 samples; 0.2 and 0.8 m): P. scabra was found at four sites, all on the western shores, two at 0.2 m and two at 0.8 m depths. Only 8 M. costata and 8 T. jordani were found, at seven sites. In 21 of the samples, no snails were found.
April–May 2010 (19 sites; 55 samples; 0.2, 0.8 and 8 m): Live T. scabra were abundant at nine sites at the 0.2 and 0.8 m depths. A number of M. costata and T. jordani were found on stones and boulders at 12 sites and a single M. tuberculata was found. At 8 m P. scabra was found at all 19 sites mostly at densities of hundreds of individuals (up to 700) per 1,000 cm³ sediment. At this depth, only nine M. tuberculata and no other native snails were found.
July 2010 (5 sites; 25 samples; 0.2, 0.8, 5, 8 and 12 m depth, hereafter all depths): P. scabra was the most abundant snail at all sites, in densities of hundreds and thousands of individuals per 1,000 cm³ sediment, with maximum abundance of 6,400 at 5 m depth at the Gofra site, on the eastern shore. At 8 m depth the density dropped to several hundred per 1,000 cm³ sediment and at 12 m it reached only a few dozens. The native T. jordani, M. costata, M. tuberculata and B. phialensis were found at some of the sites in negligible numbers; in one 0.8 m sample, however, 200 T. jordani were recorded, constituting 15 % of the total number of snails in the sample. At another site, 38 M. tuberculata were found in 1,000 cm³ sediment, this being the largest number of this species recorded in a single sample during the entire study.
November 2010 (19 sites; 81 samples; all depths; Fig. 2): P. scabra was found in 78 of the samples with high numbers at depth, including up to 7,000 per 1,000 cm³ sediment at 5 m. In the shallow water the densities were lower, usually hundreds per 1,000 cm³ sediment. T. jordani and M. costata were found on stones and boulders in shallow water; at two sites (each with two samples) these two species constituted the majority of snails in the samples and in another four sites over 25 % of the snails. M. tuberculata was found in ten of the samples, only at 0.2 and 0.8 m, at densities of up to 12 snails per 1,000 cm³ sediment. During this survey 111,649 P. scabra were collected. However, only 5,579 native snails were collected, of which 95 % were T. jordani. There were thus 20 times more individuals of a single alien species than of all native species put together.
March 2011 (19 sites; 86 samples; all depths; Fig. 2). In the shallow water, no live snails were found at most sites. In contrast, many live snails (thousands per 1,000 cm3 sediment) were found in samples from 5, 8 or 12 m, where P. scabra constituted >90 % of the total.
May 2011 (5 sites; 25 samples; all depths). In the shallow water, live snails (all species except M. tuberculata) were rare, with the total number of snails ranging from 0 to 42 per 1,000 cm3 sediment. In the deeper depths, P. scabra was the dominant species at all sites and depths, reaching hundreds (12 m) to thousands (5 and 8 m) of snails per 1,000 cm3 sediment, whereas the native fauna was represented by T. jordani only, in very low numbers (4–12 in several 8 m samples).
July 2011 (5 sites; 24 samples; all depths). P. scabra was by far the most frequent species, in all but one of the samples, where T. jordani was more frequent.
November 2011 (6 sites; 28 samples; all depths). At one site at 0.2 m, no live snails were found. P. scabra was the dominant snail in all other samples, averaging ~80 % of the total snails sampled at the shallow depths, and 99–100 % at the deeper depths (5, 8, 12 m). The abundance of P. scabra was highly variable, ranging from 0 to >4,000 individuals per 1,000 cm3 sediment. No M. tuberculata were found in this survey.
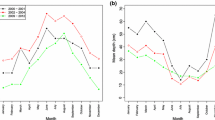
Tracking the abundance of P. scabra at two sites over the entire study period illustrated temporal patterns in their distribution (Fig. 3). At both sites, located in the south-eastern (Shittim) and north-western (Ginosar) portions of the lake, the snails disappeared from the shallow depths in March, returning in the summer, while maintaining high densities throughout the year at the deeper depths. This annual pattern was observed in both 2010 and 2011.
Temporal changes in Pseudoplotia scabra abundance (numbers of live individuals per 1,000 cm3, sediment, logarithmic scale) at different depths at a Shittim beach (south-eastern shore of lake), b Ginnosar beach (north-western shore of lake). Due to the log scale, densities of 0 individuals are presented as 0.1
Discussion
The earliest record of P. scabra in the lake was in late 2007 (Mienis and Mienis 2008). By January 2010 empty shells were found at eight sites, all on the southern shores. It is unclear whether the shells represent snails that lived and died on the spot or were carried by wave action and cast onto these shores from more distant sites. Empty P. scabra shells in the shoreline shell deposits corresponding to different water levels may indicate when the species began to spread. In the south-eastern part of the lake the shores slope moderately, so even a small rise in water level floods a large area. On this shore, there were no P. scabra shells in the shoreline shell deposits formed during the high water level of April 2004 (−208.87 m above mean sea level). The presence of a number of empty shells 3–4 m towards the lake from this waterline, coinciding with the March 2005 high water mark (the highest water level for that year: −209.94 m above mean sea level) suggest this as the year when the snails first became established in the lake. It is not known how P. scabra was initially introduced into Lake Kinneret but it is possible that it was by aquatic birds migrating from Africa and Arabia to Euro-Asia, since this species is today found throughout the Arabian Peninsula (Neubert 1998) and in eastern Africa.
In February 2010 live P. scabra individuals were found at only two sites, a month later at four sites, and after another month they were already found in the shallow and deep (8 m) water at eight sites. By summer 2010 P. scabra was present at all depths and sites sampled and in large densities. By the fall of that year the populations already consisted of thousands of individuals per 1,000 cm3 sediment. Thus, our study captured the rapid dispersal and population explosion of this invasive species. By the end of 2010, P. scabra dominated the snail community, nearly displacing the native species; this situation was maintained throughout 2011.
A key factor that may have contributed to the rapid spread of P. scabra is that when it first invaded Lake Kinneret, the native fauna was impoverished, as noted by our 2004 survey, and that the numbers of native snails were low in the early 2010 surveys before the wide dispersal of P. scabra. Indeed M. tuberculata, once the most abundant snail in soft-sediment shores of Lake Kinneret, was rarely found in our 2010–2011 surveys. M. costata, collected by the thousands in the 1980s, has also become much less abundant. While such a comparison is largely qualitative because of differences in sampling methods, the striking differences between then and now are noteworthy.
Additional factors contributing to the rapid spread of P. scabra include the tendency of young individuals to float by hanging from the water film and passively drift to new shores, its flexibility in inhabiting both soft and hard substrates, its ability to reproduce asexually so that a single individual can colonise a new site, and its viviparous mode of reproduction, which precludes egg predation. In an in situ experiment in Lake Kinneret, 35 individuals placed in a cage in the shallow water increased to 1,806 individuals in 6 months, i.e. ~50 offspring per individual. Thus, high fecundity was probably an additional contributing factor.
The invasion of P. scabra occurred some 12 years after major ecological changes in the plankton of Lake Kinneret (Zohary 2004; Roelke et al. 2007; Gal and Anderson 2010): in the mid-1990s two species of nitrogen fixing cyanobacteria and a filamentous chlorophyte invaded the lake and established bloom-forming populations; and in parallel, spring dinoflagellate blooms, previously occurring every year, did not develop in 10 of the 16 years between 1996 and 2012. These changes were attributed in part to increased amplitude of water level fluctuations, far beyond the natural amplitude of ~1.5 m, due to excessive extraction of water for human use (Zohary and Ostrovsky 2011). Currently however, we know far too little regarding the impact of these factors on the snail fauna. We do not understand how changes in the plankton, or increased water level fluctuations, could have caused a decline of the native detritus-feeding snail fauna on the one hand, yet success of an invasive detritus-feeding snail on the other.
There still are many springs and brooks throughout Israel in which the fauna is entirely native. However, just 30 km south of Lake Kinneret, in springs of the Bet Shean Valley, another alien thiarid, Tarebia granifera, has already been recorded, also originating in South-east Asia and also reproducing asexually and by viviparity (Ben-Ami 2006). This invasion by another thiarid does not portend a bright future for the native aquatic snails of Israel. These two (and other) alien invasive species may disperse throughout many of Israel’s aquatic ecosystems, displacing the native species and gradually driving them to local extinction. The aquatic habitats of Israel may well become a homogeneous system of invasive species, as in other parts of the world (Cowie 2001).
Israel is not the only place in which the native snail fauna is being displaced by alien species. In the Pacific Islands there was once an immense variety of terrestrial snails, alongside a lesser variety of freshwater ones. In recent decades this variety has rapidly diminished as species native to the islands have given way, and continue to give way, to a much smaller group of tropical widespread species that are easily dispersed by humans (Cowie 2000, 2001). Widespread and abundant invasive species in the Pacific islands include the thiarids reported in this paper: M. tuberculata, P. scabra and T. granifera (some authors consider many of the Pacific island thiarid species to be native, though not these three). In Africa, Genner et al. (2004, 2008) reported on an invasion, into Lake Malawi, of a form of Melanoides tuberculata of South-east Asian origin. This invasive form is resistant to indigenous trematodes in the lake, which castrate and induce gigantism in native M. tuberculata, thus gaining advantage in terms of reproductive capacity and survivorship. Another alien freshwater snail, Potamopyrgus antipodarum Gray (Hydrobiidae), a native of New Zealand, has successfully spread through Europe, Australia, North America and Asia since 1859, assisted by its high reproduction rate (six generations per year, with one individual producing an average of 230 juveniles per annum) and ability to survive a wide range of salinities and temperatures (Alonso and Castro-Díez 2008).
Worldwide, the two major factors responsible for loss of biodiversity are the ruin of natural habitats and the conquests by alien species. Today, these two factors seem to be endangering also the aquatic snail fauna of Israel.
References
Alonso A, Castro-Díez P (2008) What explains the invading success of the aquatic mud snail Potamopyrgus antipodarum (Hydrobiidae, Mollusca)? Hydrobiologia 614:107–116
Ben-Ami F (2006) First report of the invasive freshwater snail Tarebia granifera (Lamarck, 1816) (Gastropoda: Thiaridae) from Israel. Nautilus 120:156–161
Cowie RH (2000) Non-indigenous land and freshwater mollusks in the islands of the Pacific: conservation impacts and threats. In: Sherley G (ed) Invasive species in The Pacific: a technical review and regional strategy. South Pacific Regional Environment Programme, Apia, pp 143–172
Cowie RH (2001) Invertebrate invasions on Pacific islands and the replacement of unique native faunas: a synthesis of land and freshwater snails. Biol Invasions 3:119–136
Dagan D (1972) The influence of light and salinity on snails of the genus Theodoxus in Israel. PhD, thesis, the Hebrew University of Jerusalem. (Hebrew)
Gal G, Anderson W (2010) A novel approach to detecting a regime shift in a lake ecosystem. Methods Ecol Evol 1:45–52
Genner MJ, Michel E, Erpenbeck D, de Voogd N, Witte F, Pointier J-P (2004) Camouflaged invasion of Lake Malawi by an oriental gastropod. Mol Ecol 13:2135–2141
Genner MJ, Michel E, Todd J (2008) Resistance of an invasive gastropod to an indigenous trematode parasite in Lake Malawi. Biol Invasions 10:41–49
Glaubrecht M, Brinkmann N, Poppe J (2009) Diversity and disparity ‘down under’: systematics, biogeography and reproductive modes of the ‘marsupial’ freshwater Thiaridae (Caenogastropoda, Cerithiodea) in Australia. Zoosystemat Evol 85:199–275
Heller J (2010) Land snails of the land of Israel. Pensoft, Sofia
Mienis HK (2012) What is the correct generic name of the invasive tropical thiarid species occurring in Israel and elsewhere that was described originally as Buccinum scabrum Müller, 1774? Ellipsaria 14:14–16
Mienis HK, Mienis D (2008) Thiara scabra, a tropical snail, has invaded the Sea of Galilee, Israel. Triton 18:35–36
Neubert E (1998) Annotated checklist of the freshwater and terrestrial molluscs of the Arabian Peninsula with descriptions of new species. Fauna of Arabia 17:333–461
Roelke DL, Zohary T, Hambright KD, Montoya JV (2007) Alternative states in the phytoplankton of Lake Kinneret, Israel (Sea of Galilee). Freshwater Biol 52(3):399–411
Tchernov E (1975) The molluscs of the Sea of Galilee. Malacologia 15:147–184
Zohary T (2004) Changes to the phytoplankton assemblage of Lake Kinneret after decades of a predictable, repetitive pattern. Freshwat Biol 49:1355–1371
Zohary T, Ostrovsky I (2011) Ecological impacts of excessive water level fluctuations in stratified freshwater lake. Inland Waters 1:47–59
Acknowledgments
This project was funded by Igud Arim Kinneret.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heller, J., Dolev, A., Zohary, T. et al. Invasion dynamics of the snail Pseudoplotia scabra in Lake Kinneret. Biol Invasions 16, 7–12 (2014). https://doi.org/10.1007/s10530-013-0500-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-013-0500-5