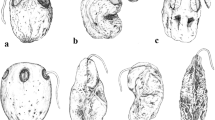
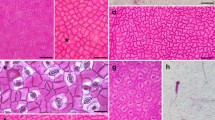
Abstract
Exocarp anatomy of 30 species of Atraphaxis has been studied to shed light on possible diagnostic and phylogenetic significance of carpological characters. The diversity of the exocarp structure observed in Atraphaxis was comparable to that in the entire tribe Polygoneae. The size of the exocarp cells, the size and shape of the lumen and its branching were the most variable. The radial size of the exocarp cells and the thickening of the walls determined the size and shape of the lumen in the exocarp cells of ripe fruits. The occurrence of the types of the exocarp cells among species of Atraphaxis only partly confirmed an earlier morphology-based division of the genus into three sections. Optimizations of the evolution of exocarp characters using parsimony on plastid phylogeny of Atraphaxis showed homoplastic changes in many characters and the absence of subclades with unique synapomorphies. Some new species identified using plastid data were confirmed by exocarp characters. Species with thick exocarp are distributed mainly in mountain regions of Central Asia, whereas species with thin exocarp are widespread in steppes and semi-deserts of Eurasia. Fruit coloration associated with the amount of phenolic compounds in the exocarp cells might be related to differences in the timing of seed germination. Species with light-brown fruits are widespread, while those with dark or black fruits are mostly local endemics.








Similar content being viewed by others
References
Aleshina LA, Lovelius OL, Sjabrjaj SV (1978) Explorationes morphologiae pollinis specierum generis Atraphaxis L. florae URSS. In: Klokov M (ed) Novosti sistematiki vysshikh i nizshikh rasteniy (Novitates systematicae plantarum vascularium et non vascularium 1977). Naukova dumka, Kiev, pp 108–122
Baasanmunkh S, Urgamal M, Oyuntsetseg B, Sukhorukov AP, Tsegmed Z, Son DC, Erst A, Oyundelger K, Kechaykin AA, Norris J, Kosachev P, Ma J-S, Chang KS, Choi HJ (2022) Flora of Mongolia: annotated checklist of native vascular plants. PhytoKeys 192:63–169. https://doi.org/10.3897/phytokeys.192.79702
Bao B, Grabovskaya-Borodina AE (2003) Atraphaxis L. In: Wu ZY, Raven PH, Hong DY (eds) Flora of China 5. Science Press, Beijing, pp 328–332
Bentham G, Hooker JD (1880) Polygonaceae. In: Bentham G, Hooker JD (eds) Genera plantarum ad exemplaria imprimis in herbariis Kewensibus servata definita 3(1). Reeve & Co, London, pp 88–105
Boissier E (1879) Flora Orientalis sive enumeratio plantarum in Oriente a Graecia et Aegypto ad Indiae fines hucusque observatorum 4. Apud. H. Georg Bibliopolam, Lugduni, Genevae et Basel.
Borodina AE (1989) Polygonaceae. In: Borodina AE, Grubov VI, Grudzinskaja IA, Menitsky JL (eds) Plantae Asiae Centralis 9. Nauka, Leningrad, pp 77–129
Brandbyge J (1993) Polygonaceae. In: Kubitzki K, Rohwer JG, Bittrich V (eds) The families and genera of vascular plants 2. Springer, Berlin, pp 531–544. https://doi.org/10.1007/978-3-662-02899-5_63
Cullen J (1967) Atraphaxis L. In: Davis PH (ed) Flora of Turkey and the East Aegean islands 2. Edinburgh University Press, Edinburgh, pp 266–267
Dammer U (1893) Polygonaceae. In: Engler A, Prantl K (eds) Die natürlichen Pflanzenfamilien, T. 3, Abt. 1a. Engelmann, Leipzig, pp 1–36
Fedotova TA (1991) Polygonaceae. In: Takhtajan A (ed) Anatomia seminum comparativa. T. 3. Nauka, Leningrad, pp 83–94
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.2307/2408678
Geiger H, Fuggerera H (1979) Über den Chemismus der Wiesner-Reaktion auf Lignin. Z Naturf b 34:1471–1472. https://doi.org/10.1515/znb-1979-1028
GenBank (2022) GenBank®. National Library of Medicine, Bethesda. Available at: https://www.ncbi.nlm.nih.gov/genbank/
Ghimire B, Choi GE, Jeong MJ, Lee H, Lee KM, Son SW, Lee CH, Suh GU (2016) Fruit morphology and anatomy of Persicaria chinensis (Polygonaceae). J Jap Bot 91:218–225
Graham SA, Wood CE (1965) The genera of Polygonaceae in the South Eastern United States. J Arnold Arbor 46:91–121
Gubanov IA (1996) Conspectus of flora of Outer Mongolia: vascular plants. Valang, Moscow
Hall TA (1999) BioEdit: a user-friendly biological sequence editor alignment and analysis program for Windows 95/98/NT. Nucl Acids Symp Ser 41:95–98
Hatfield R, Vermerris W (2001) Lignin formation in plants. The dilemma of linkage specificity. Pl Physiol 126:1351–1357. https://doi.org/10.1104/pp.126.4.1351
Hendry GAF, Thompson K, Moss CJ, Edwards E, Thorpe PC (1994) Seed persistence: a correlation between seed longevity in the soil and ortho–dihydroxyphenol concentration. Funct Ecol 8:658–664. https://doi.org/10.2307/2389929
Huang G, McCrate AJ, Varriano-Marston E, Paulsen GM (1983) Caryopsis structural and imbibitional characteristics of some hard red and white wheats. Cereal Chem 60:161–165
Ivlev VI (2008) Introduction of rare and disappearing plants in Central Kazakhstan. Bot Res Sib Kaz (Bot Issl Sibiri Kazakhstana) 14:79–89
Jaubert H–FC, Spach E (1844–1846) Illustrationes Plantarum Orientalium 2. Roret, Paris. Tabs. 101–200
Katoh K, Misawa K, Kuma K, Miyata T (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucl Acids Res 30:3059–3066. https://doi.org/10.1093/nar/gkf436
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Molec Biol Evol 30:772–780. https://doi.org/10.1093/molbev/mst010
Khan M, Cavers PB, Kane M, Thompson K (1997) Role of the pigmented seed coat of proso millet (Panicum miliaceum L.) in imbibition, germination and seed persistence. Seed Sci Res 7:21–26. https://doi.org/10.1017/S0960258500003329
Kong MJ, Hong SP (2018) Comparative achene morphology of Persicaria sect. Cephalophilon and related taxa (Polygonaceae). Korean J Pl Taxon 48:134–142. https://doi.org/10.11110/kjpt.2018.48.2.134
Kostina MV, Yurtseva OV (2021) Structure and developmental rhythm of shoot systems of A. frutescens (L.) K. Koch., A. replicata Lam., and A. pyrifolia Bunge (Atraphaxis L., Polygonaceae). Contemp Probl Ecol 14:241–254. https://doi.org/10.1134/S1995425521030070
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Molec Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lattanzio V, Lattanzio VM, Cardinali A (2006) Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. Phytochem Adv Res 661:23–67
Lattanzio V, Kroon PA, Quideau S, Treutter D (2008) Plant phenolics—secondary metabolites with diverse functions. In: Daayf F, Lattanzio V (eds) Recent advances in polyphenol research. Blackwell, Chichester, pp 1–35. https://doi.org/10.1002/9781444302400.ch1
Lewis NG, Yamamoto E (1990) Lignin: occurrence, biogenesis and biodegradation. Annual Rev Pl Biol 41:455–496. https://doi.org/10.1146/annurev.pp.41.060190.002323
Li X, Zhao X, Wang Z, Dong Z (2010) Seed dormancy-breaking and germination requirements of two Atraphaxis species. Bull Bot Res 30:600–603. https://doi.org/10.7525/j.issn.1673-5102.2010.05.014
Li X, Zhao X, Yu R (2011) Effects of seed maturation time and dry storage on germination of two Atraphaxis species. Acta Ecol Sin 30:3727–3732
Lonay H. 1922. Génèse et anatomie des péricarpes et des spermodermes chez les Polygonaceés. Mém Soc Roy Sci Liége, Sér. 3, Tome 2, Part 2, 6: 1–109
Lovelius OL (1978) Compositio specierum, Distributio geographica et cohaerentia oecologica generis Atraphaxis L. (Polygonaceae). In: Klokov MV (ed) Novosti sistematiki vysshikh i nizshikh rasteniy (Novitates systematicae plantarum vascularium et non vascularium 1977). Naukova dumka, Kiev, pp 85–108
Lovelius OL (1979) Synopsis generis Atraphaxis L. (Polygonaceae). Novosti Sist Vyssh Rast 15:114–128
Marek S (1954) Cechy morfologiczne i anatomiczne owoców rodzajów Polygonum L. i Rumex L. oraz klucze do ich oznaczania (Morphological and anatomical features of the fruits of genera Polygonum L., Rumex L. and keys for their determination). Monogr Bot 2:77–161. https://doi.org/10.5586/mb.1954.002
Matilla A, Gallardo M, Puga-Hermida MI (2005) Structural, physiological and molecular aspects of heterogeneity in seeds: a review. Seed Sci Res 15:63–76. https://doi.org/10.1079/SSR2005203
Meisner CF (1857) Polygonaceae. In: de Candolle AP (ed) Prodromus systematis naturalis regni vegetabilis 14. Masson, Paris, pp 1–186
Miroslavov EA (1974) Struktura i funktsiya epidermy lista pokrytosemennykh (The structure and function of the epidermis of the angiosperm leaf). Nauka, Leningrad
Mohamed-Yasseen Y, Barringer SA, Splittstoesser WE, Costanza S (1994) The role of seed coats in seed viability. Bot Rev 60:426–439. https://doi.org/10.1007/BF02857926
Nei M, Kumar S (2000) Molecular evolution and phylogenetics. Oxford University Press, New York
Nixon KC (2002) WinClada, version 1.00.08. Computer program published and distributed by the author, Ithaca. Available at: http://www.cladistics.com
Pavlov VN (1970) Atraphaxis L. In: Komarov VL (ed) Flora of the USSR (Flora SSSR) 5. Izdatel'stvo Akademii Nauk SSSR, Moskva & Leningrad, 1936, pp 392–411. [Translated from Russian by Dr Landau N, Israel Program for Scientific Translations, Jerusalem 1970]
Patzlaff A, McInnis S, Courtenay A, Surman C, Newman LJ, Smith C, Bevan MW, Mansfield Sh, Whetten RW, Sederoff RR, Campbell MM (2003) Characterisation of a pine MYB that regulates lignification. Pl J 36:743–754. https://doi.org/10.1046/j.1365-313X.2003.01916.x
Powell AA (1989) The importance of genetically determined seed coat characteristics to seed quality in grain legumes. Ann Bot (Oxford) 63:169–175. https://doi.org/10.1093/oxfordjournals.aob.a087720
Puga-Hermida MI, Gallardo M, del Carmen R-G, Matilla AJ (2003) The heterogeneity of turnip–tops (Brassica rapa) seeds inside the silique affects germination, the activity of the final step of the ethylene pathway, and abscisic acid and polyamine content. Funct Pl Biol 30:767–775. https://doi.org/10.1071/FP03053
Rechinger KH, Schiman-Czeika M (1968) Polygonaceae. In: Rechinger KH (ed) Flora Iranica 56/28. Academishe Druck-u.Verlagsanstalt, Graz, pp 1–88
Ronse De Craene LP, Hong SP, Smets E (2000) Systematic significance of fruit morphology and anatomy in tribes Persicarieae and Polygoneae (Polygonaceae). Bot J Linn Soc 134:301–337. https://doi.org/10.1111/j.1095-8339.2000.tb02356.x
Roth I (1977) Fruits of angiosperms. Handbuch der Pflanzenanatomie 10(1), 2nd edn. Borntraeger, Berlin
Royal Botanic Gardens Kew (2021) Seed Information Database (SID). Version 7.1. Available at: http://data.kew.org/sid/
Schatral A, Osborne JM (2006) Germination and dormancy states of seeds in the environmental weed Rumex vesicarius (Polygonaceae). I. Seed polymorphism and germination of extracted seeds. Austral J Bot 54:773–782. https://doi.org/10.1071/BT04201
Schuster TM, Reveal JL, Kron KA (2011) Phylogeny of Polygoneae (Polygonaceae: Polygonoideae). Taxon 60:1653–1666. https://doi.org/10.1002/tax.606010
Sirrine EF (1894) Structure of seed coats of Polygonaceae. Proc Iowa Acad Sci 2:128–135
Tavakkoli S, Kazempour Osaloo Sh, Mozaffarian V, Maassoumi A (2015) Molecular phylogeny of Atraphaxis and the woody Polygonum species (Polygonaceae): taxonomic implications based on molecular and morphological evidence. Pl Syst Evol 301:1157–1170. https://doi.org/10.1007/s00606-014-1140-7
Thiers B. (2021, continuously updated) Index Herbariorum: a global directory of public herbaria and associated staff. New York Botanical Garden’s virtual herbarium, Bronx. Available at: http://sweetgum.nybg.org/science/ih/. Accessed 18 Oct 2021
Webb DA (1993) Atraphaxis L. In: Tutin TG, Burges NA, Chater AO, Edmonson JR, Heywood VH, Moore DM, Valentine DH, Walters SM, Webb DA (eds) Flora Europaea 1, 2nd edn. Cambridge University Press, Cambridge, p 108
Yurtseva OV (2001) Ultrasculpture of achene surface in Polygonum section Polygonum (Polygonaceae) in Russia. Nordic J Bot 21:513–528. https://doi.org/10.1111/j.1756-1051.2001.tb00805.x
Yurtseva OV, Severova EE, Bovina IYu (2014) Pollen morphology and taxonomy of Atraphaxis (Polygoneae, Polygonaceae). Pl Syst Evol 300:749–766. https://doi.org/10.1007/s00606-013-0917-4
Yurtseva OV, Kuznetsova OI, Mavrodieva ME, Mavrodiev EV (2016a) What is Atraphaxis L. (Polygonaceae, Polygoneae): cryptic taxa and resolved taxonomic complexity instead of the formal lumping and the lack of morphological synapomorhies. PeerJ 4:1–52. https://doi.org/10.7717/peerj.1977
Yurtseva OV, Kuznetsova OI, Mavrodiev EV (2016b) A broadly sampled 3–loci plastid phylogeny of Atraphaxis (Polygoneae, Polygonoideae, Polygonaceae) reveals new taxa: I. Atraphaxis kamelinii spec. nov. from Mongolia. Phytotaxa 268:1–24. https://doi.org/10.11646/phytotaxa.268.1.1
Yurtseva OV, Severova EE, Mavrodiev EV (2017) Persepolium (Polygoneae): a new genus in Polygonaceae based on conventional maximum parsimony and three-taxon statement analyses of a comprehensive morphological dataset. Phytotaxa 314:151–194. https://doi.org/10.11646/phytotaxa.314.2.1
Yurtseva OV, Mavrodiev EV (2019) Caelestium genus novum (Polygonaceae, Polygoneae): evidence based on the results of molecular phylogenetic analyses of tribe Polygoneae, established with consideration of the secondary structure of the ITS rDNA regions. Novosti Sist Vyssh Rast 50:47–79. https://doi.org/10.31111/novitates/2019.50.47
Yurtseva OV, Lazkov GA, Ukrainskaja UA, Deviatov AG (2019) Caelestium (Polygonaceae, Polygoneae), evidence based on morphology. Novosti Sist Vyssh Rast 50:80–100. https://doi.org/10.31111/novitates/2019.50.80
Yurtseva OV, Badmaeva NK, Mavrodiev EV (2021a) A broadly sampled 3-loci plastid phylogeny of Atraphaxis (Polygoneae, Polygonoideae, Polygonaceae) reveals new taxa: II. Atraphaxis selengensis spec. nov. and A. davurica Jaub. et Spach from Russian Transbaikalia. Phytotaxa 484:44–74. https://doi.org/10.11646/phytotaxa.484.1.2
Yurtseva OV, Severova EE, Deviatov AG, Olonova MV, Samigullin TH (2021b) Polygonum schischkinii is a member of Atraphaxis (Polygonaceae, Polygoneae): evidences from morphological and molecular analyses. Phytotaxa 491:193–216. https://doi.org/10.11646/phytotaxa.491.3.1
Yurtseva OV, Vasilieva NV, Kostikova VA, Samigullin TH (2022) A broadly sampled 3-loci plastid phylogeny of Atraphaxis (Polygoneae, Polygonoideae, Polygonaceae) reveals new taxa: III. Atraphaxis kuvaevii and cryptic species in Atraphaxis pungens (M.Bieb.) & Spach from Southern Siberia and Northern Mongolia. Phytotaxa (in press)
Zhang XX, Xiong XH, Xu F, Deng LN, Wang KM, Hou YT (2011) Exocarp anatomy of Polygonum and related species from Shandong Province. Pl Sci J 529:554–551
Zhang M-L, Sanderson SC, Sun Y-X, Byalt VV, Hao X-L (2014) Tertiary montane origin of the Central Asian flora: evidence inferred from cpDNA sequences of Atraphaxis (Polygonaceae). J Integr Pl Biol 56:1125–1135. https://doi.org/10.1111/jipb.12226
Acknowledgements
We thank O.V. Cherneva, A.E. Borodina, V.I. Dorofeev, A.P. Seregin, N.M. Reshetnikova, G.A. Lazkov, A.I. Shmakov, A.N. Kupriyanov, E.V. Banaev, V.V. Chepinoga and O.A. Anenkhonov for their kind permission to take samples for morphological investigations from Herbaria LE, MW, MHA, FRU, ALTB, KUZ, NS, NSK, IRKU and UUH. We are grateful to M.G.Pimenov, N.S. Gamova, S. Svirin and N.K. Badmaeva who collected some specimens used in this study. We thank A.C. Timonin for valuable comments that helped to improve the text. We thank anonymous Reviewers for their attentive attitude, helpful comments and advice.
Funding
This work was supported by the Russian Foundation for Basic Research, project no. 20-04-00033.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any research involving humans or animals as research objects.
Additional information
Handling Editor: Ferhat Celep.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Information on Electronic Supplementary Material
Information on Electronic Supplementary Material
Online resource 1. The origin of the material used for anatomical and molecular study. Left: Species names, the numbers of fruit samples and the voucher information (herbarium acronyms according to Index Herbariorum) for samples used in anatomical study of fruits. Right: DNA sample numbers and the voucher information for samples used in molecular studies, and GenBank numbers of sequences. In some cases, the same samples were used in both analyses, but in other cases, different samples were studied. “—” no data.
Online resource 2. The combined aligned plastid matrix for cpDNA trnL intron + trnL-F IGS and rpl32-trnL (UAG) IGS regions including 54 nucleotide sequences for accessions of Atraphaxis (52), Persepolium (1), and Bactria (1).
Online resource 3. Morphological characters of fruit and exocarp and their states used in Maximum Parsimony analyses.
Online resource 4. Morphological data matrix used in Maximum Parsimony analyses. “0” and “1” are character states, “ ? ” denotes unknown or ambiguous character state.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yurtseva, O.V., Deviatov, A.G. & Sokoloff, D.D. Exocarp structure in the genus Atraphaxis (Polygonaceae, Polygoneae). Plant Syst Evol 308, 32 (2022). https://doi.org/10.1007/s00606-022-01824-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00606-022-01824-0